Understanding Glycolysis: The Central Pathway of Carbohydrate Metabolism
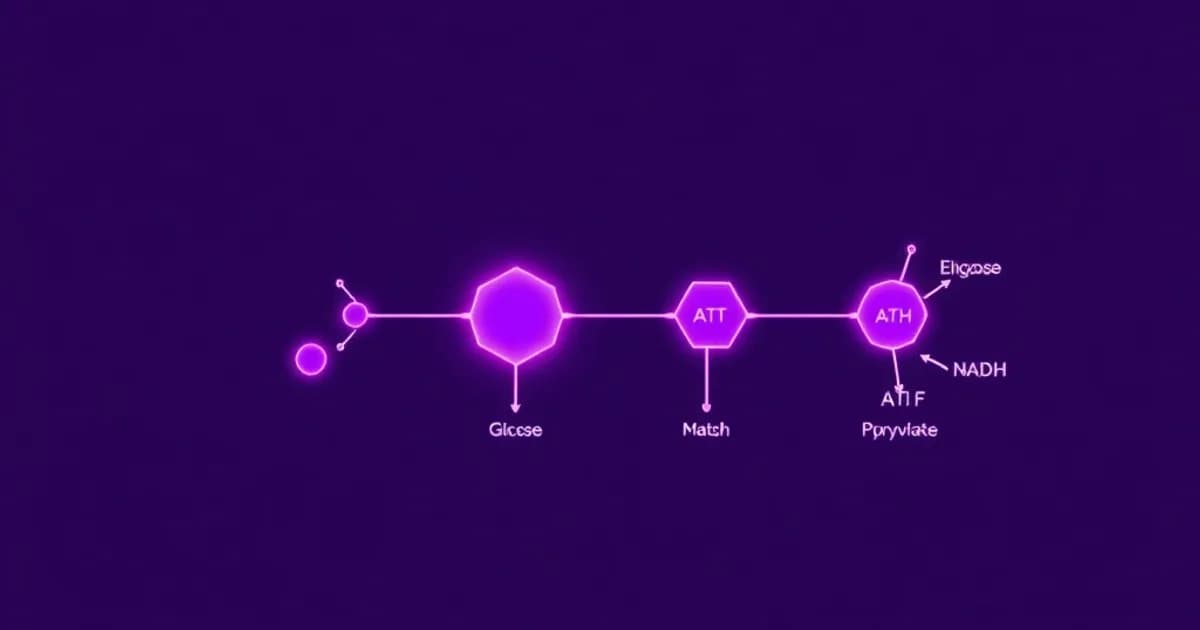
Glycolysis is the metabolic pathway that converts glucose into pyruvate in your cell's cytoplasm. This ten-step process is fundamental to cellular energy production and connects dietary carbohydrates to major metabolic pathways like the citric acid cycle, lipogenesis, and amino acid synthesis.
The Two Phases of Glycolysis
Glycolysis divides into two distinct phases. The preparatory phase (steps 1-3) requires ATP investment to modify glucose. The payoff phase (steps 6-10) generates ATP and NADH through substrate-level phosphorylation.
Each glucose molecule generates a net of two ATP and two NADH per glucose under anaerobic conditions. This makes glycolysis the only energy-producing pathway available when oxygen is limited.
How Glucose Enters the Pathway
Glucose enters your cell through GLUT proteins and is immediately phosphorylated to glucose-6-phosphate by hexokinase. This phosphorylation step is crucial because it traps glucose inside the cell and commits it to metabolism.
Steps 4 and 5 reorganize the carbon skeleton without significant energy changes. Steps 6-10 generate ATP through substrate-level phosphorylation, where high-energy phosphate compounds transfer directly to ADP to form ATP.
Why Understanding Flow Matters
Recognizing each individual step matters, but recognizing the overall flow and regulation distinguishes high-scoring MCAT students from average ones. Focus on how the pathway connects as one integrated system, not isolated reactions.
Key Enzymes and Regulatory Points in Glycolysis
Three enzymes in glycolysis serve as irreversible steps and major regulatory points: hexokinase, phosphofructokinase (PFK), and pyruvate kinase. Understanding how each enzyme is regulated determines whether glycolysis speeds up or slows down.
Phosphofructokinase: The Master Control Switch
Phosphofructokinase (PFK) is widely considered the most important regulatory enzyme in glycolysis. PFK catalyzes the first committed step, converting fructose-6-phosphate to fructose-1,6-bisphosphate.
PFK is subject to multiple allosteric regulations. It is inhibited by ATP and citrate (signals of abundant energy) and activated by AMP, ADP, and fructose-2,6-bisphosphate (signals of low energy and fed state).
Hexokinase and Pyruvate Kinase Regulation
Hexokinase catalyzes the first step and is inhibited by its product, glucose-6-phosphate, providing feedback regulation. Pyruvate kinase, the final enzyme, is inhibited by ATP and alanine while being activated by fructose-1,6-bisphosphate through feed-forward activation.
Other Important Enzymes
Phosphoglycerate kinase and pyruvate kinase catalyze substrate-level phosphorylation, the mechanism generating ATP during glycolysis. Aldolase splits fructose-1,6-bisphosphate into two three-carbon molecules, allowing cells to utilize non-hexose sugars.
Lactate dehydrogenase (LDH) is not technically part of glycolysis but is essential for anaerobic metabolism. LDH regenerates NAD+ from NADH when oxygen is unavailable, allowing glycolysis to continue in anaerobic conditions.
Energy Yield and Cofactor Requirements in Carbohydrate Metabolism
The energy yield from glycolysis depends on the cell type and metabolic state. Under anaerobic conditions, glycolysis produces a net of two ATP and two NADH per glucose molecule.
Where ATP Comes From
ATP is generated through substrate-level phosphorylation during steps 7 and 10, catalyzed by phosphoglycerate kinase and pyruvate kinase respectively. Since glucose splits into two three-carbon units after step 4, each glucose produces two molecules of each product.
NADH and the Electron Transport Chain
The NADH produced at step 6 must be recycled back to NAD+ for glycolysis to continue. In anaerobic conditions, lactate dehydrogenase regenerates NAD+. In aerobic conditions, NADH enters the mitochondrial electron transport chain.
When NADH enters the electron transport chain aerobically, each NADH generates approximately 2.5 ATP molecules. Combined with the citric acid cycle, total ATP from one glucose reaches approximately 7-8 ATP. The MCAT often simplifies this to approximately 30-32 ATP per glucose including all cellular respiration stages.
Critical ATP and Phosphate Requirements
Two ATP molecules are consumed in the preparatory phase (by hexokinase and phosphofructokinase), representing the initial ATP investment. Inorganic phosphate is required at step 6 for the glyceraldehyde-3-phosphate dehydrogenase reaction, making phosphate availability essential for pathway continuation.
Regulation by Metabolic State: Fed, Fasted, and Exercise States
Carbohydrate metabolism is tightly regulated depending on your body's metabolic state, a concept tested frequently on MCAT biochemistry. Each state activates or suppresses different enzymes to match your current energy needs.
Fed State: Glucose Abundance
In the fed state (after eating), glucose is abundant and insulin levels elevate. Insulin promotes glucose uptake into cells and activates phosphofructokinase through elevated F-2,6-BP levels.
Under fed state conditions, glycolysis proceeds at high rates. Excess glucose converts to glycogen (glycogenesis) or fat (lipogenesis), favoring ATP production and biosynthetic pathways.
Fasted State: Energy Mobilization
In the fasted state, blood glucose is maintained through gluconeogenesis and glycogenolysis, not glycolysis. Glucagon elevates, which decreases F-2,6-BP levels and inhibits glycolysis while promoting gluconeogenesis.
The fasted state represents a metabolic shift toward energy mobilization from stored sources rather than immediate glucose utilization.
Exercise State: Increased Demands
During exercise, muscles have increased ATP demands, causing AMP and ADP levels to rise. This activates phosphofructokinase and increases glycolytic flux to meet energy needs.
The Pasteur effect describes the increase in glycolytic rate when anaerobic conditions emerge. Anaerobic conditions produce less ATP per glucose, requiring cells to process more glucose to meet energy demands.
The Cori Cycle Connection
The Cori cycle links muscle glycolysis to hepatic gluconeogenesis. Lactate produced by muscles during intense exercise transports to the liver, where it converts back to glucose through gluconeogenesis and returns to muscles.
Clinical Relevance and Common Genetic Disorders Affecting Carbohydrate Metabolism
Several genetic disorders affecting glycolysis and carbohydrate metabolism appear in MCAT biochemistry courses. Understanding these conditions demonstrates why biochemistry matters beyond exam preparation.
Key Genetic Disorders
- Pyruvate dehydrogenase deficiency results in lactate and alanine accumulation, as pyruvate cannot convert efficiently to acetyl-CoA.
- Phosphofructokinase deficiency impairs glycolytic flux despite normal glucose uptake.
- Von Gierke disease (glucose-6-phosphatase deficiency) prevents the final step of both gluconeogenesis and glycogenolysis, leading to severe hypoglycemia and lactic acidosis.
Metabolic Dysfunction in Disease
Lactate accumulation is a key clinical sign in many metabolic disorders. Understanding the lactate shuttle (Cori cycle) is essential for interpreting these conditions.
Hyperglycemia, whether from diabetes mellitus or other causes, impairs metabolic pathways through the Randle cycle (glucose-fatty acid cycle), where elevated glucose oxidation suppresses fatty acid oxidation.
Cancer Cells and the Warburg Effect
The Warburg effect is reversed in tumor cells, where cancer cells preferentially utilize glycolysis even in the presence of oxygen, producing lactate at high rates. This metabolic shift is partly explained by rapid ATP generation from glycolysis and production of biosynthetic precursors needed for rapid cell division.
Lactic Acidosis
Lactic acidosis can result from type A causes (tissue hypoxia) or type B causes (mitochondrial dysfunction, malignancy, liver disease). Recognizing these clinical connections helps you appreciate biochemistry's medical relevance and improves retention through clinically relevant examples.