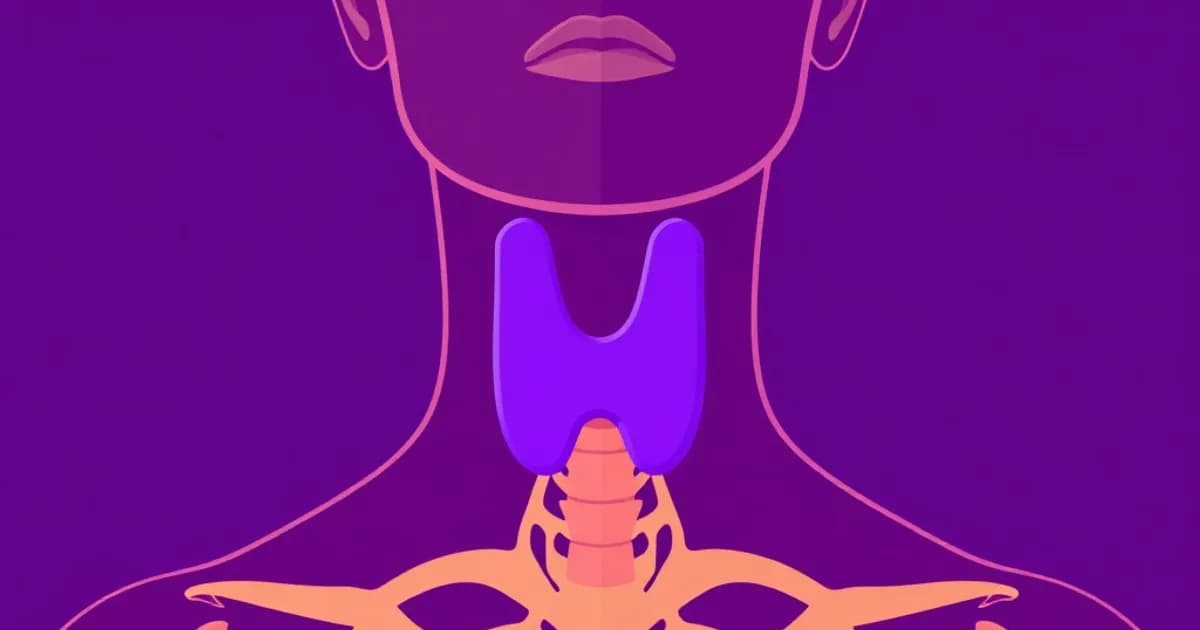
Gross Anatomy and Location of the Thyroid Gland
Position and Shape
The thyroid gland sits in your anterior neck, positioned just below the thyroid cartilage (Adam's apple). It extends from the second tracheal ring to the fourth tracheal ring. The gland has a distinctive butterfly shape with two lateral lobes connected by a central isthmus.
In adults, the thyroid weighs approximately 15 to 20 grams. The right lobe is typically larger than the left, measuring about 4 to 6 cm in length and 2 to 3 cm in width.
Capsule and Internal Structure
A fibrous capsule derived from cervical fascia encloses the gland. This capsule contains extensions called septa that divide the gland into small lobules. Deep to the thyroid lie critical structures: the trachea, esophagus, and recurrent laryngeal nerve.
Damage to the recurrent laryngeal nerve during surgery can cause voice changes or hoarseness. This anatomical relationship is clinically significant for any thyroid procedure.
Blood Supply and Drainage
The thyroid receives rich blood supply from:
- Superior thyroid arteries (branches of the external carotid)
- Inferior thyroid arteries (branches of the thyrocervical trunk)
The extensive blood supply reflects the gland's high metabolic activity. Venous drainage occurs through the superior and middle thyroid veins into the internal jugular vein, with inferior thyroid veins draining into the brachiocephalic veins.
Knowing these anatomical relationships is critical for surgery, clinical exams, and understanding how structural problems affect nearby organs.
Histological Structure and Cell Types
Thyroid Follicles
Thyroid follicles are the functional units of the gland. Each follicle consists of a single layer of follicular cells (also called thyrocytes) surrounding a central cavity. This cavity contains colloid, a protein-rich substance filled with thyroglobulin.
Follicular cells are simple cuboidal epithelial cells that synthesize, store, and secrete thyroid hormones. They produce T3 (triiodothyronine) and T4 (thyroxine).
Follicle Activity Levels
Follicle appearance changes based on activity level. Active follicles appear smaller with cuboidal cells and minimal colloid storage. Inactive follicles are larger with flattened cells and abundant colloid.
This visual difference reflects how hard the follicle is working to produce hormones.
Parafollicular Cells and Connective Tissue
Parafollicular cells (C cells) are scattered among follicles and produce calcitonin, a hormone involved in calcium regulation. Unlike follicular cells derived from the endoderm, C cells originate from the neural crest.
Connective tissue between follicles carries blood vessels, lymphatic vessels, and nerves. Clusters of undifferentiated cells in this tissue can develop into new follicles as needed.
This histological organization explains how the thyroid produces hormones and how diseases like thyroiditis or thyroid cancer disrupt normal function.
Thyroid Hormone Synthesis and Secretion
The Iodine and Tyrosine Foundation
Thyroid hormone synthesis requires iodine, tyrosine, and multiple enzymatic steps. The process begins when iodide ions are actively transported into follicular cells through a sodium-iodide symporter on the cell membrane.
Once inside, iodide is oxidized to iodine by thyroid peroxidase (TPO). Simultaneously, tyrosine residues within thyroglobulin are iodinated in the colloid.
Formation of T3 and T4
Iodination creates two intermediates:
- Monoiodotyrosine (MIT) contains one iodine atom
- Diiodotyrosine (DIT) contains two iodine atoms
TPO couples these together to form:
- T4 (one MIT plus one DIT)
- T3 (two DITs)
These hormones remain stored in colloid as inactive precursor until your body needs them.
Release into the Bloodstream
When the body requires thyroid hormones, follicular cells take up colloid through pinocytosis. Lysosomal enzymes break down thyroglobulin and release free T3 and T4 into the bloodstream.
T4 accounts for about 80% of thyroid secretion, while T3 represents about 20%. However, T3 is more biologically active. Both hormones travel bound to thyroid hormone-binding proteins in blood, with only a small free fraction being physiologically active.
TSH Regulation
Thyroid-stimulating hormone (TSH) from the pituitary controls hormone production. TSH binds to follicular cell receptors and increases all synthesis, storage, and release steps. This pathway is essential for understanding hyperthyroidism, hypothyroidism, and antithyroid medications.
Hormonal Functions and Metabolic Effects
Metabolic Rate and Energy Use
Thyroid hormones affect virtually every tissue in your body. Their primary function is increasing the basal metabolic rate (energy used at rest). T3 and T4 enter cells and bind to thyroid hormone receptors in the nucleus, activating genes for metabolic enzymes.
These hormones increase oxygen consumption, heat production, and the breakdown of carbohydrates, fats, and proteins.
Cardiovascular and Nervous System Effects
Thyroid hormones increase heart rate and cardiac output. They enhance nervous system responsiveness and improve muscle strength and tone.
They also influence mood, cognition, and temperature regulation in adults.
Development and Growth
Thyroid hormones are critical for normal skeletal and neurological development in infants and children. Severe deficiency during development can cause permanent intellectual disability.
This is why newborn screening programs test for congenital hypothyroidism.
Calcitonin and Calcium Regulation
The gland produces calcitonin from parafollicular cells, which lowers blood calcium by inhibiting bone resorption and promoting kidney excretion. Calcitonin's physiological role is minor compared to parathyroid hormone and vitamin D.
Feedback Loop Control
Thyroid hormone levels maintain themselves through negative feedback. Elevated T3 and T4 suppress TSH from the anterior pituitary. Low thyroid hormone levels stimulate TSH secretion.
This feedback loop keeps hormone levels within a narrow optimal range. Clinical assessment measures TSH, free T4, and sometimes free T3 to diagnose thyroid disorders.
Clinical Significance and Common Pathologies
Hypothyroidism
Hypothyroidism is insufficient thyroid hormone production. It results from thyroid dysfunction (primary), pituitary or hypothalamic disease (secondary), or iodine deficiency.
Symptoms include fatigue, weight gain, cold intolerance, and slowed metabolism. Treatment uses levothyroxine thyroid hormone replacement.
Hyperthyroidism
Hyperthyroidism is excessive thyroid hormone production. Graves' disease, an autoimmune condition, is the most common cause. Toxic nodules also cause hyperthyroidism.
Symptoms include rapid heart rate, heat intolerance, anxiety, and weight loss. Treatments include antithyroid drugs, radioactive iodine, or surgery.
Thyroid Cancer and Nodules
Thyroid cancer includes papillary, follicular, medullary, and anaplastic types. It originates from follicular cells or parafollicular cells and requires surgical removal and sometimes radioactive iodine therapy.
Thyroid nodules are extremely common and usually benign but require evaluation to rule out malignancy.
Goiter and Thyroiditis
Goiter is thyroid enlargement occurring with hypothyroidism, hyperthyroidism, or normal hormone levels. Causes include iodine deficiency, autoimmune disease, or benign nodules.
Thyroiditis is gland inflammation. Hashimoto's disease destroys follicular cells through autoimmunity. Subacute thyroiditis involves viral inflammation.
Surgical Risks
Knowing thyroid anatomy is crucial for surgery. Damage to the recurrent laryngeal nerve causes hoarseness or voice loss. The parathyroid glands embedded in the thyroid capsule may be inadvertently damaged, leading to hypoparathyroidism and low blood calcium.