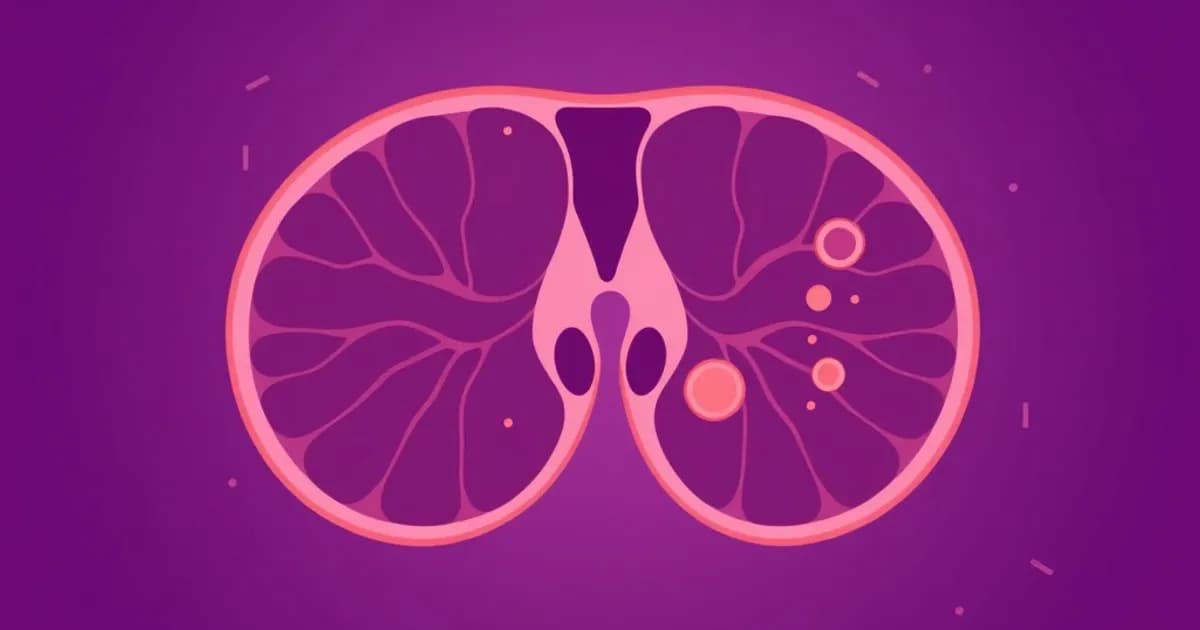
Gross Anatomy and Location of the Thymus
The thymus is a bilobed organ positioned in the anterior mediastinum, directly behind the sternum. It extends from the second rib down to the fourth rib in adults. A fibrous capsule encloses each lobe and sends dividers deep into the organ, creating distinct lobules.
Size and Vascular Supply
The thymus reaches maximum weight at puberty (30-40 grams) then gradually shrinks. Infants and children have proportionally larger thymic tissue that may extend into the neck. Internal thoracic arteries and thyroid arteries supply rich blood flow to this highly vascular organ.
Clinical and Imaging Importance
The thymus's position behind the sternum protects it but makes it visible on chest imaging. Proximity to major blood vessels requires careful surgical consideration if thymic pathology needs intervention. You should identify the thymus on cross-sectional imaging and recognize how its size changes throughout life.
Microscopic Organization and Histological Structure
Each thymic lobule divides into an outer cortex and inner medulla. The cortex appears dark (basophilic) due to densely packed developing lymphocytes called thymocytes. Epithelial reticular cells form a support network and produce cytokines essential for T cell development.
The Medulla and Hassall's Corpuscles
The medulla contains fewer lymphocytes but features unique Hassall's corpuscles - concentrically arranged keratinized epithelial cells found nowhere else. These structures help eliminate autoreactive T cells and promote regulatory T cell (Treg) development. They produce thymic stromal lymphopoietin, which influences T cell differentiation.
The Blood-Thymus Barrier
The blood-thymus barrier protects developing T cells from systemic antigens. It consists of the thymic capsule, perivascular spaces, and endothelial cells. This barrier is critical during the vulnerable selection phases of T cell maturation.
Supporting Stromal Cells
The thymic stroma contains dendritic cells, macrophages, and medullary epithelial cells. These present self-antigens for negative selection, teaching T cells which targets to ignore. Understanding this architecture explains how positive and negative selection work.
T Cell Development and Selection Processes
Thymopoiesis is the process of T cell development and maturation in the thymus. Lymphoid progenitor cells migrate from bone marrow into the thymus where they undergo dramatic transformation and selection.
Positive Selection in the Cortex
Developing T cells start in the subcapsular cortex as double-negative cells (lacking CD4 and CD8 markers). As they move toward the medulla, they express both markers, becoming double-positive cells. During positive selection, T cells must recognize self-MHC molecules presented by cortical epithelial cells. This ensures only T cells with functional receptors survive. T cells failing this interaction undergo apoptosis.
Negative Selection in the Medulla
Approximately 95 percent of developing thymocytes die during thymic residence. As selected T cells reach the medulla, they encounter self-antigens presented by medullary epithelial cells and professional antigen-presenting cells. T cells reacting strongly to self-antigens are eliminated, preventing autoimmune disease.
Exit as Single-Positive Cells
Only surviving single-positive T cells (expressing either CD4 or CD8, but not both) exit to populate peripheral lymphoid tissues. These cells can recognize pathogens while tolerating the body's own tissues.
Thymic Involution and Age-Related Changes
The thymus undergoes progressive involution starting after puberty, accelerating after age 40. Functional thymic tissue is replaced with adipose (fat) and fibrous tissue. In elderly individuals, the thymus becomes a small, fatty remnant in the anterior mediastinum.
Impact on Immune Function
This involution significantly contributes to immunosenescence (age-related immune deterioration). The reduced T cell output explains diminished vaccination responses and increased infection susceptibility in older adults. Despite shrinkage, the thymus retains metabolic activity and produces some T cells throughout life.
Clinical Relevance in Different Age Groups
Thymic hyperplasia in children can cause anterior mediastinal masses. In adults, thymic pathology more often involves tumors such as thymomas. Residual thymic function becomes important in patients experiencing lymphocyte depletion from chemotherapy or immunosuppressive therapy.
Measuring Thymic Function
Recent thymic emigrants are markers used to assess thymic function following bone marrow transplantation or HIV treatment with highly active antiretroviral therapy (HAART). This measurement provides insights into immune reconstitution.
Clinical Significance and Pathological Conditions
Several conditions affect the thymus and demonstrate its critical immune function role.
Thymic Tumors and Associated Conditions
Thymomas and thymic carcinomas are primary thymic neoplasms presenting with chest pain, cough, or superior vena cava syndrome. These tumors frequently associate with myasthenia gravis, an autoimmune condition affecting neuromuscular junctions. Thymectomy (thymus removal) is sometimes performed therapeutically for myasthenia gravis.
Thymic Hyperplasia and Hypoplasia
Thymic hyperplasia (enlargement) occurs with severe infection or chronic antigenic stimulation. Thymic hypoplasia or aplasia (congenital deficiency) occurs in DiGeorge syndrome (22q11 deletion), causing severe T cell immunodeficiency. DiGeorge syndrome presents on a spectrum from partial to complete thymic aplasia.
Diagnostic and Clinical Recognition
Radiologic recognition of normal thymic tissue versus pathological enlargement or tumors is essential for proper diagnosis. Thymic disorders can present with diverse symptoms ranging from immunodeficiency to autoimmunity. Any compromise to thymic function affects both cellular and humoral immunity downstream. Understanding these relationships helps predict clinical presentation.