Mechanism of Action and Renal Physiology
How SGLT2 Inhibitors Work in the Kidney
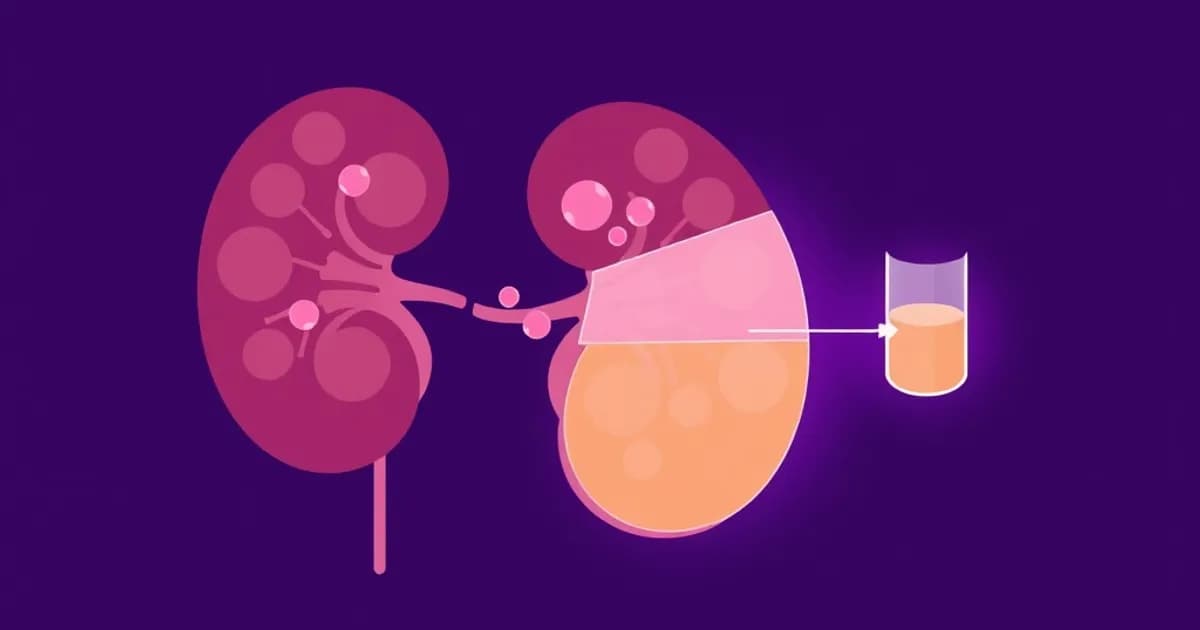
SGLT2 inhibitors work by blocking sodium-glucose cotransporter 2 proteins. These proteins are located in the S1 segment of the proximal convoluted tubule in the nephron. In normal physiology, glucose filtered at the glomerulus is completely reabsorbed in the proximal tubule via SGLT1 and SGLT2 transporters.
SGLT2 accounts for approximately 90 percent of glucose reabsorption. SGLT1 handles the remaining 10 percent. When empagliflozin binds to and inhibits SGLT2, glucose cannot be reabsorbed. Instead, it is excreted in the urine, resulting in glycosuria and reduced blood glucose levels.
Glucose-Dependent Mechanism and Safety Benefits
This mechanism is glucose-dependent, meaning hypoglycemia risk is minimal compared to insulin secretagogues. The drug reduces plasma glucose by approximately 25 to 30 mg/dL. HbA1c drops by 0.5 to 1.5 percent in monotherapy.
Beyond glucose excretion, SGLT2 inhibition produces important metabolic effects. These include natriuresis, mild osmotic diuresis, and weight reduction of 2 to 3 kilograms. Empagliflozin is highly selective for SGLT2 over SGLT1, which is crucial because SGLT1 inhibition in the gut would cause gastrointestinal adverse effects.
Insulin-Independent Glucose Lowering
The drug exhibits insulin-independent glucose lowering. This makes it effective across the entire spectrum of type 2 diabetes severity. It is compatible with virtually all other antidiabetic agents without requiring additional insulin secretion.
Pharmacokinetics, Dosing, and Administration
Standard Dosing and Administration
Empagliflozin is administered orally as a tablet available in 10 mg and 25 mg formulations. The standard starting dose is 10 mg once daily in the morning. You may increase to 25 mg daily for enhanced glycemic control.
Peak plasma concentrations occur within 1.5 to 2 hours of oral administration. Food has no clinically significant effect on absorption, so timing with meals does not matter.
Absorption and Metabolism
The drug exhibits approximately 86 percent oral bioavailability. Its plasma half-life is 12.4 hours, allowing once-daily dosing. Empagliflozin undergoes hepatic metabolism via glucuronidation, mediated primarily by UDP-glucuronosyltransferase enzymes.
Minimal involvement of cytochrome P450 pathways results in minimal drug-drug interactions compared to other antidiabetic agents. Renal clearance accounts for approximately 50 percent of total drug elimination.
Renal Function and Contraindications
No dose adjustment is required for mild to moderate renal impairment. However, the drug is contraindicated in advanced chronic kidney disease with eGFR less than 20 mL/min/1.73 m2. Sufficient glomerular filtration is necessary for the drug's glucose-lowering effect.
Steady-state plasma concentrations are achieved within 5 days of initiation. Empagliflozin reaches approximately 99 percent plasma protein binding. Its volume of distribution is 73.8 liters. Due to its glucose-dependent mechanism, the drug can be administered regardless of meal timing.
Cardiovascular and Renal Protective Effects
Landmark Cardiovascular Outcomes Trial
Beyond glucose lowering, empagliflozin demonstrates remarkable cardiovascular and renal protective benefits. These have revolutionized its clinical importance. The EMPA-REG OUTCOME trial was a landmark cardiovascular outcomes study.
It demonstrated that empagliflozin reduces major adverse cardiovascular events by 14 percent. It reduces cardiovascular mortality by 38 percent. It reduces hospitalization for heart failure by 35 percent in patients with type 2 diabetes and established cardiovascular disease.
These benefits occurred independent of glycemic control. This suggests mechanisms beyond glucose reduction alone.
Chronic Kidney Disease Protection
The EMPA-KIDNEY trial demonstrated that empagliflozin slows chronic kidney disease progression. It reduces cardiovascular events in patients with CKD regardless of diabetes status. Renal protective mechanisms include reduction of intraglomerular pressure through afferent arteriolar vasodilation.
Decreased tubular sodium reabsorption reduces tubuloglomerular feedback. Anti-inflammatory and antifibrotic effects contribute to kidney protection. Weight reduction and blood pressure lowering provide additional cardiovascular benefits. Systolic blood pressure reductions of 3 to 4 mmHg are observed.
The drug reduces albuminuria by approximately 30 to 40 percent.
Heart Failure Benefits
Heart failure benefits include improved cardiac efficiency through metabolic shift. The heart uses ketone and lactate utilization more efficiently. Reduced cardiac preload occurs through natriuresis. Direct cardioprotective effects add additional benefit.
These pleiotropic effects have expanded empagliflozin use beyond type 2 diabetes. It is now used in chronic kidney disease and heart failure management. This makes it increasingly central to chronic disease management across multiple specialties. It represents a fundamental shift in antidiabetic therapy paradigms.
Adverse Effects, Drug Interactions, and Clinical Contraindications
Common and Genital Adverse Effects
Empagliflozin is generally well-tolerated, but several important adverse effects require careful clinical attention. Genital mycotic infections occur in 10 to 15 percent of patients, particularly women. Glycosuria creates favorable conditions for fungal growth. Urinary tract infections may occur with increased frequency.
Volume Depletion and Serious Risks
Osmotic diuresis can cause intravascular volume depletion. This is particularly problematic in patients on loop diuretics or with compromised renal function. This may lead to hypotension and acute kidney injury.
Euglycemic diabetic ketoacidosis is rare with incidence less than 0.1 percent, but represents a serious concern. Ketoacidosis develops at glucose levels less than 250 mg/dL, making diagnosis challenging. Risk increases with acute illness, surgery, or insulin discontinuation.
Additional Safety Concerns
Amputation risk has been debated in literature. The CANVAS trial noted increased lower extremity amputation risk, though this finding has not been consistently replicated. Bone fracture risk may be slightly increased due to calciuria-induced mineral loss.
Volume depletion contraindications include severe renal impairment, type 1 diabetes, and acute decompensated heart failure. Drug interactions are minimal due to glucuronidation metabolism. However, inducers of UDP-glucuronosyltransferase might reduce empagliflozin efficacy.
Drug Interactions and Special Populations
Concurrent use with other antidiabetic agents requires dose adjustment to prevent hypoglycemia. The drug is contraindicated in pregnancy and lactation. Elderly patients and those with significant comorbidities require careful dose adjustment and monitoring.
Clinical Efficacy and Role in Diabetes Management Algorithms
Position in Modern Treatment Guidelines
Empagliflozin has established itself as a preferred agent in modern diabetes management algorithms. The American Diabetes Association and European Association for the Study of Diabetes consensus guidelines recommend it prominently. As monotherapy, empagliflozin reduces HbA1c by 0.5 to 1.5 percent depending on baseline glycemic control and dosing.
When combined with other agents, additive glycemic control is observed with minimal hypoglycemia risk. The glucose-dependent mechanism minimizes this risk.
Use in Cardiovascular and Kidney Disease
The drug is particularly valuable for patients with type 2 diabetes and established cardiovascular disease. Cardioprotective benefits provide rationale for use independent of glycemic targets. It is recommended for diabetic patients with chronic kidney disease or albuminuria due to renal protective effects.
Weight reduction of 2 to 3 kilograms is consistent and clinically meaningful. This addresses a common concern in diabetic populations. Blood pressure reduction through natriuresis and osmotic effects provides additional cardiovascular benefit.
Combination Therapy and Real-World Effectiveness
Empagliflozin can be used as first-line agent in newly diagnosed type 2 diabetes or added to existing regimens. Combination therapy with metformin, GLP-1 receptor agonists, DPP-4 inhibitors, sulfonylureas, and insulin is well-established. These have complementary mechanisms.
The drug maintains efficacy across eGFR ranges down to 20 mL/min/1.73 m2, though caution is necessary with advanced renal disease. Real-world effectiveness studies demonstrate approximately 70 to 80 percent of patients achieve their individualized glycemic targets within 12 weeks. Sustained benefit occurs over years of therapy.
Cost considerations vary by insurance, but SGLT2 inhibitors are increasingly considered cost-effective given cardiovascular and renal benefits.