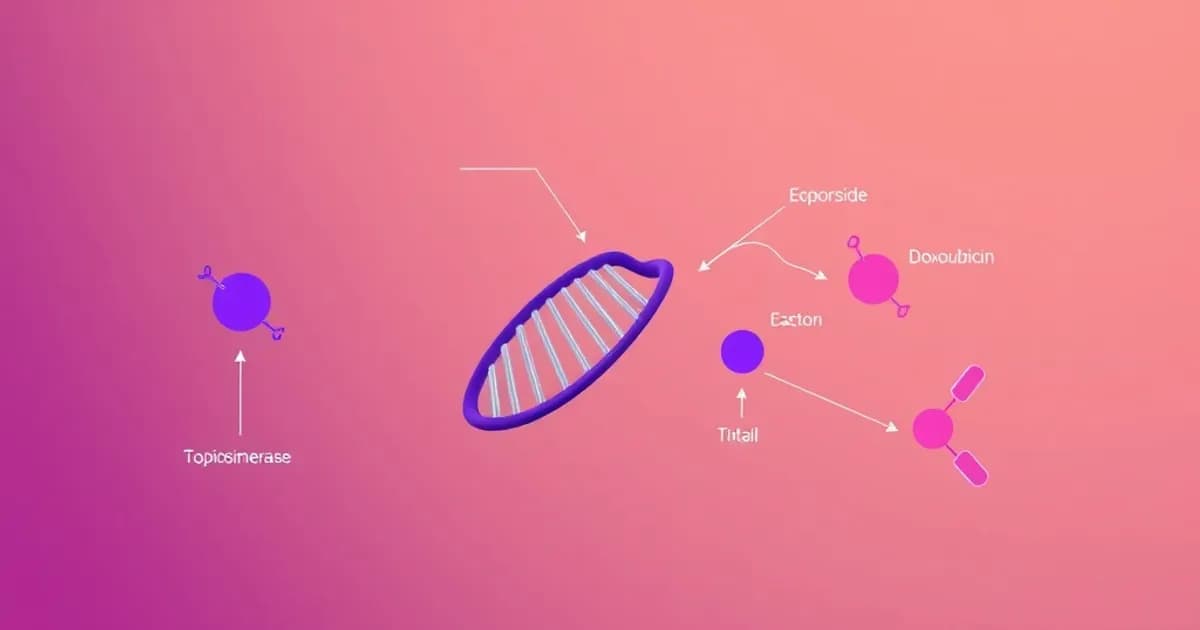
Mechanism of Action and Topoisomerase Function
Topoisomerases are enzymes that manage DNA structure by creating temporary breaks. During normal cell division, topoisomerase II breaks both DNA strands, allows them to unwind, and then reseals them.
How Etoposide and Doxorubicin Block Topoisomerase II
Both drugs stabilize the enzyme-DNA complex and prevent the resealing step. This traps the topoisomerase on the DNA strand. When replication machinery hits these stabilized complexes, irreversible double-strand breaks accumulate. Cancer cells die through apoptosis (programmed cell death) because they cannot survive this DNA damage.
Key Structural Differences
Etoposide and doxorubicin work differently at the molecular level:
- Etoposide: Non-intercalating inhibitor. Works purely through topoisomerase blocking. A semisynthetic podophyllotoxin derivative.
- Doxorubicin: Intercalating agent. Inserts between DNA base pairs while also blocking topoisomerase. Additionally generates free radicals that cause extra DNA damage.
Doxorubicin's dual action makes it more potent but also more toxic to normal cells. This explains why doxorubicin causes more serious side effects like heart damage.
Why Cancer Cells Are Most Vulnerable
Rapidly dividing cancer cells require constant DNA replication and therefore encounter topoisomerase inhibitors frequently. Normal cells divide slowly and escape much of the toxicity, making these drugs relatively selective for cancer.
Clinical Applications and Therapeutic Uses
Etoposide and doxorubicin treat different cancers based on their unique properties and how they move through the body.
Etoposide Treatment Uses
Etoposide is the primary choice for:
- Small cell lung cancer (SCLC)
- Testicular cancer
- Lymphomas and leukemias
- Often combined with other chemotherapy drugs
Etoposide can be given intravenously or taken as a pill, giving doctors flexibility in treatment planning. Good CNS penetration makes it useful for brain tumors.
Doxorubicin Treatment Uses
Doxorubicin is a cornerstone agent for:
- Breast cancer (often in AC combination with cyclophosphamide)
- Lymphomas and leukemias
- Soft tissue sarcomas
- Part of ABVD regimen for Hodgkin lymphoma
The choice between etoposide and doxorubicin depends on cancer type, prior treatments, and patient factors like heart health.
Why Combination Therapy Matters
Both drugs are rarely used alone. Combination regimens achieve synergistic effects and overcome cancer cell resistance. For example, AC-T (doxorubicin plus cyclophosphamide followed by taxanes) is standard breast cancer care. Understanding these combinations is crucial for clinical practice.
Pharmacokinetics and Drug Properties
How your body processes these drugs determines dosing schedules, monitoring needs, and which patients are at higher risk for problems.
Etoposide Pharmacokinetics
Etoposide has a half-life of 4 to 11 hours depending on route (IV versus oral). The liver metabolizes it. Oral absorption is variable (25 to 75 percent), requiring careful monitoring. It penetrates the central nervous system well, helping it reach brain tumors but also increasing neurotoxicity risk.
Doxorubicin Pharmacokinetics
Doxorubicin has a much longer half-life of 20 to 48 hours. The liver metabolizes it, but it also accumulates extensively in the heart muscle. This tissue binding explains why doxorubicin causes cumulative cardiotoxicity. It does not penetrate the brain well due to its size and water-repelling properties.
Why These Differences Matter Clinically
Doxorubicin's long half-life and heart accumulation explain why:
- Maximum lifetime dose is limited to approximately 450 mg/m2
- Cardiac monitoring with echocardiography or MUGA scans is mandatory
- Dosing intervals are typically 3 to 4 weeks to allow recovery
- Etoposide can be redosed sooner without cardiac risk
Both drugs need dose reductions in patients with liver dysfunction. Kidney problems are less critical but still matter.
Adverse Effects and Management Strategies
These powerful drugs cause serious side effects requiring prevention, monitoring, and aggressive supportive care.
Doxorubicin's Major Adverse Effects
Cardiotoxicity is the most concerning:
- Occurs in about 5 percent of patients above 450 mg/m2 cumulative dose
- Can cause congestive heart failure
- Presents acutely as arrhythmias during infusion
- Requires baseline and periodic heart monitoring
Other major effects include:
- Alopecia (hair loss) in most patients
- Extravasation tissue necrosis if IV leaks
- Increased risk of secondary leukemias years later
- Nausea and vomiting
- Myelosuppression (low blood counts)
Etoposide's Major Adverse Effects
Myelosuppression is the primary dose-limiting toxicity, particularly low white blood cells and platelets. Effects typically recover within 2 to 3 weeks. Other effects include:
- Peripheral neuropathy at higher doses
- Ototoxicity (hearing loss)
- Secondary leukemia risk
- Nausea and vomiting
Managing These Effects
Prevention and monitoring are key:
- Use 5-HT3 antagonists and NK1 antagonists to prevent nausea
- Give G-CSF support for severe myelosuppression
- Monitor liver function at baseline and periodically
- Use dexrazoxane for doxorubicin cardioprotection in high-risk patients
- Consider scalp cooling to reduce hair loss
- Check baseline heart function and recheck regularly for doxorubicin
- Administer through central lines when possible to prevent extravasation
Drug Interactions and Clinical Considerations
These drugs interact with many medications through shared metabolic pathways, potentially increasing toxicity or reducing effectiveness.
Cytochrome P450 Interactions
Both etoposide and doxorubicin are metabolized by CYP3A4 and CYP2C9 enzymes. Medications that inhibit these enzymes increase drug levels:
- Cyclosporine significantly increases etoposide levels and toxicity
- Azole antifungals (fluconazole, itraconazole) inhibit metabolism
- Macrolide antibiotics (erythromycin) increase drug levels
Medications that induce these enzymes decrease drug levels:
- St. John's Wort reduces etoposide and doxorubicin effectiveness
- Rifampin dramatically reduces drug levels
Cardiotoxicity Interactions with Doxorubicin
Combining doxorubicin with other cardiotoxic drugs substantially increases heart failure risk:
- Trastuzumab (HER2 inhibitor)
- Tyrosine kinase inhibitors
- Radiation to the chest
- Calcium channel blockers may increase doxorubicin toxicity
P-Glycoprotein and Drug Efflux
P-glycoprotein transporters pump drugs out of cancer cells. Inducers of P-glycoprotein reduce drug effectiveness. This mechanism decreases intracellular drug concentration in tumor cells.
Hepatic Impairment Dosing
Liver dysfunction is more important than kidney dysfunction for dosing. Dose reduction guidelines:
- Doxorubicin: 50 percent reduction for bilirubin 1.2 to 3 mg/dL, 75 percent for above 3 mg/dL
- Etoposide: Substantial reductions also needed
- Child-Pugh scoring helps guide modifications in cirrhotic patients
Always check liver function tests before giving these drugs.