Understanding Aortic Dissection: Definition and Mechanism
How Aortic Dissection Develops
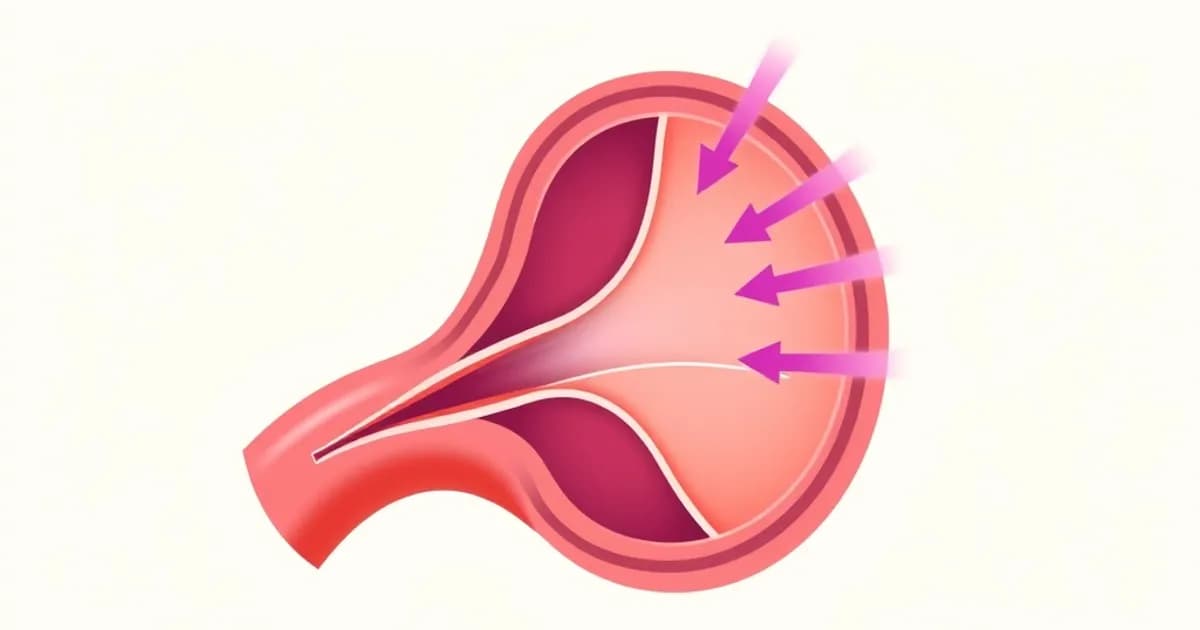
Aortic dissection occurs when blood separates the aortic wall layers, typically entering the media and creating a false lumen parallel to the true one. An intimal tear allows high-pressure blood to exploit weakness in the medial layer. This tear serves as the initiating event that starts a self-perpetuating process.
Once blood enters the false lumen, it can remain patent, thrombose, or rupture based on tear size and pressure dynamics. Understanding this mechanism explains why blood pressure control is the first priority in acute management.
The Three Aortic Layers
The aorta has three distinct layers: the intima (inner), media (muscular middle), and adventitia (outer connective tissue). A dissection specifically involves separation at or within the media. This distinguishes it from other aortic conditions like atherosclerotic aneurysms or intramural hematomas.
Why Location and Extent Matter
The dissection's specific location and extent determine treatment options. The relationship between true and false lumens affects whether branch vessels receive adequate blood flow. Controlling hemodynamic stress prevents further dissection propagation.
Risk Factors and Pathophysiological Drivers
Hypertension: The Most Common Risk Factor
Hypertension appears in 60 to 90 percent of dissection cases and is the most modifiable risk factor. Chronic elevated blood pressure causes medial degeneration through repeated wall stress. Smooth muscle cells become hypertrophied, weakening the aorta's structural integrity.
Acute severe hypertensive episodes can precipitate dissection by overwhelming the wall's capacity. This is why beta-blockers and vasodilators form the first-line treatment strategy.
Connective Tissue Disorders and Medial Weakness
Connective tissue disorders like Marfan syndrome, Ehlers-Danlos syndrome, and Turner syndrome predispose to dissection. These conditions cause abnormal collagen and elastin composition, making the aortic wall inherently fragile.
Cystic medial necrosis is a pathological process involving loss of elastic fibers and smooth muscle cells in the media. Bicuspid aortic valve disease is associated with this condition.
Other Contributing Risk Factors
Multiple other factors compromise aortic wall integrity or increase hemodynamic stress:
- Atherosclerosis affecting intimal health
- Pregnancy (particularly third trimester and peripartum)
- Cocaine use causing acute hypertension
- Chest trauma
- Age-related changes including reduced elastin and increased collagen cross-linking
Acute triggering events include severe hypertensive episodes, intense Valsalva maneuvers, or aortic procedures. Understanding these factors explains why dissection presentations vary widely.
Classification Systems: Stanford and DeBakey
The Stanford Classification
The Stanford system divides dissections into two categories based on ascending aorta involvement. This system is widely used because it directly guides treatment decisions.
Type A dissections involve the ascending aorta and represent surgical emergencies. They carry risks of rupture, tamponade, aortic regurgitation, and coronary artery compromise.
Type B dissections are limited to the descending thoracic aorta distal to the left subclavian artery. These are typically managed medically unless complications develop.
The DeBakey Classification
The DeBakey system provides more anatomical detail with three types:
- Type I involves the entire aorta from ascending to descending portions
- Type II is limited to the ascending aorta
- Type III affects only the descending thoracic aorta, subdivided into Type IIIa (above diaphragm) and Type IIIb (below diaphragm)
Clinical Implications and Outcomes
Approximately 60 to 70 percent of acute dissections are Type A, carrying higher mortality without intervention. Type A dissections require urgent surgery, while Type B typically allows medical management initially.
Chronic dissections are defined as those present for more than two weeks. They follow similar classification but have different management considerations. Imaging modality selection depends on suspected classification type, and anatomical extent impacts decisions about endovascular versus open surgical repair.
Pathophysiological Complications and Organ Malperfusion
Catastrophic Rupture and Tamponade
Aortic rupture, particularly into the pericardium, causes acute tamponade with cardiogenic shock. This is a catastrophic event requiring immediate intervention. The pericardial fluid accumulation restricts cardiac filling and severely compromises cardiac output.
Acute Aortic Regurgitation
Acute aortic regurgitation develops when dissection undermines the aortic valve apparatus. The incompetent valve allows severe backflow during diastole, causing acute left ventricular volume overload.
This can progress to acute pulmonary edema and cardiogenic shock within hours. Type A dissections commonly extend into the sinuses of Valsalva, where the valve is located.
Coronary Artery Involvement
Dissection can extend into coronary ostia, particularly affecting the right coronary artery. This causes myocardial infarction with chest pain that mimics primary coronary disease. Patients may have multiple simultaneous complications.
Branch Vessel Malperfusion
Malperfusion occurs through two mechanisms. Static obstruction happens when the dissection flap directly compromises a branch vessel orifice. Dynamic obstruction occurs when pressure differentials between lumens cause the flap to prolapse and intermittently obstruct vessels.
Malperfusion can affect multiple organ systems:
- Brain (cerebral involvement with stroke)
- Spinal cord (spinal ischemia)
- Abdomen (mesenteric ischemia)
- Limbs (peripheral ischemia)
The severity of complications depends on dissection location, tear propagation velocity, and which branch vessels are compromised. Understanding these complications explains why rapid diagnosis and immediate blood pressure control are critical.
Why Flashcards Are Effective for Aortic Dissection Pathophysiology
The Spaced Repetition Advantage
Aortic dissection pathophysiology presents unique learning challenges due to its anatomical, mechanical, and clinical complexity. Flashcard-based learning is particularly effective for mastering this topic.
Spaced repetition reinforces memory formation of key concepts like classification systems, risk factors, and complications. This spacing principle produces superior long-term retention compared to massed practice.
Breaking Down Interconnected Concepts
Aortic dissection involves multiple interconnected ideas: the mechanism of intimal tear, hemodynamic consequences, classification systems, anatomical variants, and clinical presentations. Flashcards break these into manageable units while maintaining the relationships between them.
This compartmentalization helps you master one concept thoroughly before connecting it to the next.
Active Recall Strengthens Clinical Reasoning
Active recall through flashcards forces deeper cognitive processing than passive reading. When you attempt to recall whether aortic regurgitation occurs in Type A dissections before checking the answer, you engage problem-solving skills essential for clinical practice.
This mental effort is precisely what builds reliable memory.
Visual Learning for Complex Anatomy
Flashcards accommodate the visual and spatial learning demands of this topic. Many concepts benefit from visual representation: the Stanford classification, intimal flap anatomy, and branch vessel involvement patterns.
Digital flashcard systems can incorporate diagrams, cross-sectional views, and anatomical illustrations alongside text.
Targeted Review of Knowledge Gaps
Flashcards enable you to create personalized decks targeting your specific weak areas. You might link risk factors to pathophysiological changes or pair classification types with management approaches.
This targeted review is more efficient than rereading textbooks or studying unfamiliar material. You focus your effort where it matters most.