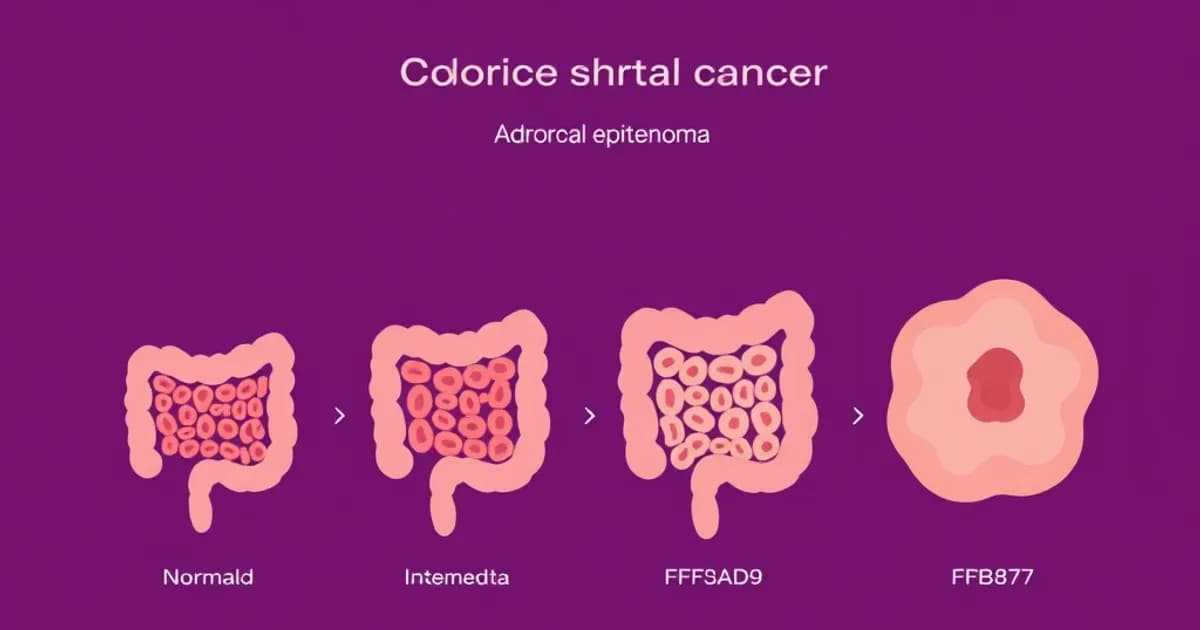
The Normal Colonic Epithelium and Early Changes
The sequence starts with normal colonic epithelium, a single layer of columnar cells organized into crypts. These cells have tight control of growth, maturation, and cell death. Any disruption of this balance opens the door to cancer.
The First Mutation: APC Gene Loss
The initiating event usually involves loss of the APC gene, a tumor suppressor that regulates the Wnt signaling pathway. When APC is lost, cells proliferate inappropriately and skip normal death signals.
This early change creates areas of increased cell density within the crypts, sometimes called dysplasia or intraepithelial neoplasia. Early detection at this stage is critical because it offers a window to prevent progression.
Why This Matters for Inherited Syndromes
People with familial adenomatous polyposis (FAP) inherit a faulty APC gene from birth. They develop hundreds of adenomas and face near-certain cancer without surgery. This proves how powerful APC mutations are in starting the cancer sequence.
The normal-to-adenoma transition establishes a malignant clone that accumulates more mutations over years or decades.
Small Adenomas and the Adenoma-Carcinoma Transition
Small adenomas (less than 1 centimeter) appear during screening colonoscopy. These are classified into three types based on appearance.
Three Adenoma Types and Their Risk Levels
- Tubular adenomas: Most common, lower malignant potential
- Tubulovillous adenomas: Intermediate pattern and risk
- Villous adenomas: Highest malignant potential
During this stage, KRAS mutations develop in about 40-50% of adenomas. KRAS promotes growth through the mitogen-activated protein kinase pathway.
Small Adenomas Can Be Stopped Here
At this point, adenomas are confined to the mucosa and submucosa. Removing them completely prevents cancer development. Most adenomas grow slowly and never progress to cancer. However, certain features signal higher risk of progression.
Distinguishing Dysplasia Levels
Pathologists grade dysplasia as either low-grade or high-grade based on nuclear size, architecture, and chromatin appearance.
Low-grade dysplasia shows increased nuclear size but preserved glandular structure. High-grade dysplasia demonstrates severe architectural distortion and abnormal nuclei. High-grade dysplasia means higher cancer risk but still no invasion through the muscularis mucosae.
Large Adenomas and Progression to Invasive Carcinoma
Large adenomas (greater than 1 centimeter) carry substantially higher malignant potential, especially with villous histology or high-grade dysplasia. As adenomas grow, additional mutations accumulate.
The TP53 Mutation: The Critical Turning Point
Mutations in the TP53 tumor suppressor gene typically occur in large adenomas. TP53 normally triggers cell death when DNA is damaged. Without functional TP53, cells ignore damage signals and continue dividing.
TP53 mutations often accompany high-grade dysplasia and mark the adenoma's approach to carcinoma.
Invasion: The Line That Matters Most
The defining moment in cancer development is invasion, when neoplastic cells penetrate through the muscularis mucosae into deeper layers. This distinction matters more than dysplasia grade.
Once invasion occurs, cancer can spread to lymph nodes and distant organs. The 10-15 year timeline from adenoma to carcinoma varies widely. Large adenomas with high-grade dysplasia require complete removal. If complete endoscopic removal is impossible, surgical resection may be necessary.
Why Aggressive Management Is Justified
Removing large adenomas before invasion is proven cancer prevention. This explains why adenoma detection through screening is one of medicine's most effective cancer prevention strategies.
Molecular Genetics and the Multi-Hit Hypothesis
The adenoma-carcinoma sequence follows the multi-hit hypothesis: cancer requires multiple genetic alterations accumulated over time. Colorectal cancer demonstrates this principle clearly.
The Classic Gene Sequence
The typical order of mutations progresses as follows:
- APC mutation (initiating event in normal epithelium or tiny adenomas): Deregulates Wnt signaling
- KRAS mutation (intermediate-sized adenomas): Activates oncogenes, promotes growth
- TP53 mutation (large adenomas): Blocks tumor suppression and apoptosis
Additional Alterations
Other genes like SMAD4, PIK3CA, and mismatch repair genes can also mutate. The specific order varies somewhat, but the principle of accumulating mutations remains constant.
Why This Model Explains Everything
Multiple mutations explain why adenomas grow slowly. Most never become cancer because the final critical mutations don't occur. Screening works because removing adenomas stops the process before late mutations occur.
Different cancers can use different molecular pathways, including the microsatellite instability pathway (mismatch repair gene defects) and the CpG island methylator phenotype pathway. However, the adenoma-carcinoma sequence remains the most common pathway for sporadic colorectal cancer.
Clinical Significance and Screening Implications
Understanding the adenoma-carcinoma sequence transforms how we prevent colorectal cancer. The 10-15 year progression time creates a perfect window for intervention.
Why Screening Works
Colonoscopy allows visualization of the entire colon and rectum. Adenomas are removed immediately (polypectomy), preventing malignant transformation. This directly interrupts the cancer pathway before invasion occurs.
Screening guidelines recommend starting at age 45-50 for average-risk individuals. All adenomas found should be removed.
Surveillance Intervals Based on Findings
Post-removal surveillance depends on adenoma characteristics:
- Normal baseline colonoscopy or adenomas less than 10 mm: 10-year interval
- Adenomas 10-20 mm or 3-10 small adenomas: 5-10 year interval
- Adenomas greater than 20 mm or sessile serrated lesions: 2-5 year interval
People with hereditary syndromes like familial adenomatous polyposis or Lynch syndrome require intensive surveillance due to extremely high adenoma burden and cancer risk.
Real-World Impact
Countries with widespread screening show declining colorectal cancer incidence and mortality. The adenoma-carcinoma sequence highlights why this happens: adenoma removal genuinely prevents cancer, not just treats a benign condition. This understanding helps clinicians counsel patients and helps patients understand screening's true importance.