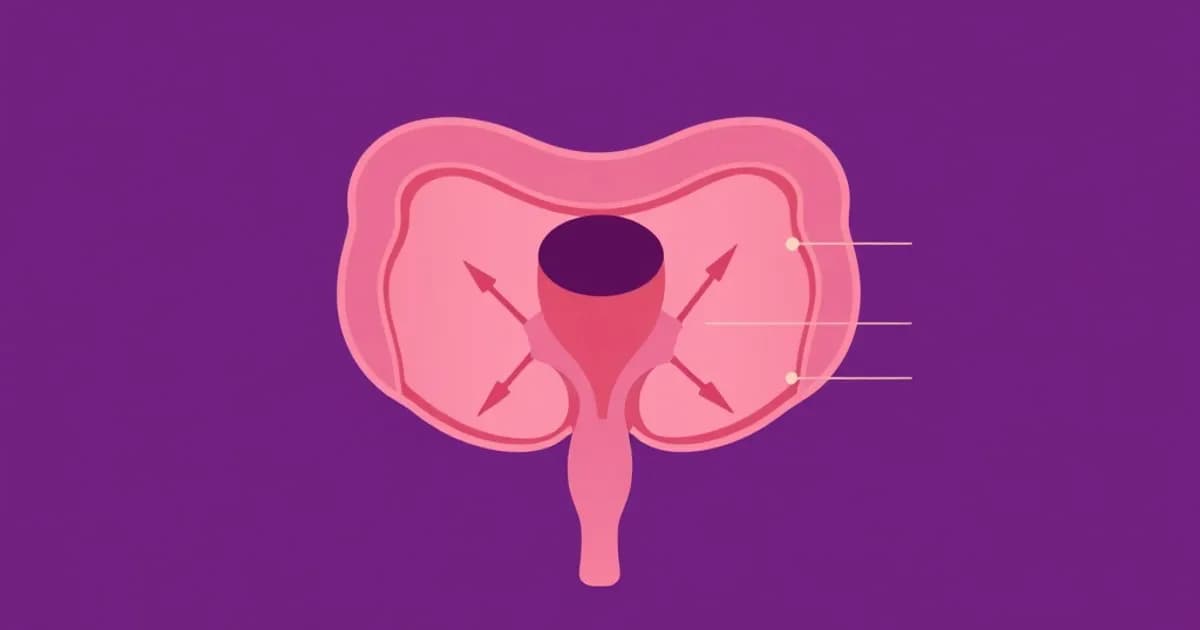
Gross Anatomy and Location of the Urinary Bladder
The urinary bladder is a retroperitoneal organ positioned in the pelvis, below the peritoneum. In males, it sits above the prostate gland and in front of the rectum. In females, it sits in front of the uterus and vagina.
How Bladder Position Changes
The bladder's position shifts based on how full it is. When empty, it looks like a small pyramidal shape. When full, it expands upward into the abdominal cavity. The bladder's apex connects to the median umbilical ligament, which is a leftover from fetal development.
The bladder base (fundus) is the lowest and most fixed part. The superior surface gets covered by peritoneum when the bladder is distended. The anterolateral walls touch the pubic bones.
Clinical Significance
Understanding these spatial relationships matters for procedures like catheterization, cystoscopy, and suprapubic aspiration. Knowing where structures are located helps surgeons access the bladder safely.
Nerve Supply
The bladder receives nerve fibers from multiple sources. Parasympathetic fibers from the pelvic splanchnic nerves (S2-S4) control bladder contractions. Sympathetic fibers from the hypogastric plexus help maintain storage. Somatic motor fibers through the pudendal nerve control your external urethral sphincter.
Knowing these connections explains why certain pathologies cause specific symptoms and why surgical approaches must account for nearby structures.
Internal Structure and Histological Layers
The bladder wall has four distinct layers that work together for filling and emptying. Each layer serves a specific function in storing and releasing urine.
The Four Bladder Layers
- Mucosa (innermost layer): Contains specialized transitional epithelium that stretches dramatically without tearing
- Submucosa: A connective tissue layer with elastic fibers and blood vessels
- Detrusor muscle: Three interlacing smooth muscle layers that contract during urination
- Serosa (outermost layer): Visceral peritoneum covering only the superior and posterolateral surfaces
Transitional Epithelium
This specialized lining is unique to the bladder. It contains dome cells that flatten and spread as pressure increases. This allows your bladder to hold 500 to 600 milliliters of urine in adults without tearing the lining.
Important Internal Structures
The internal urethral meatus marks the opening into the urethra and is surrounded by the internal urethral sphincter (smooth muscle under parasympathetic control). The trigone is a smooth triangular area on the bladder's interior base. It is bounded by the two ureteric orifices and the internal urethral meatus.
The trigone has different embryological origins than the rest of the bladder. It came from mesodermal tissue rather than endoderm. This gives it different histology and vascular supply. The extraperitoneal portions are covered by fascia instead of peritoneum.
Vascular Supply, Lymphatic Drainage, and Innervation
The bladder receives blood, drains lymph, and receives nerve signals through specialized networks. Understanding these systems helps with surgery and imaging interpretation.
Blood Supply
The superior and inferior vesical arteries provide the main blood supply to the bladder. These are branches of the anterior division of the internal iliac artery. In females, the vaginal and uterine arteries also contribute to bladder blood supply.
Vesical veins form a rich network surrounding the bladder that drains into the internal iliac veins. This vascular anatomy is important for surgical procedures and understanding bleeding patterns.
Lymphatic Drainage
Lymph from the bladder drains to internal iliac and external iliac lymph nodes. Some drainage also goes to sacral nodes. Understanding these pathways helps predict how bladder cancer spreads.
Three Innervation Pathways
Parasympathetic innervation comes from S2-S4 spinal segments via the pelvic splanchnic nerves. This provides motor control to the detrusor muscle and drives the micturition reflex.
Sympathetic innervation comes from T11-L2 via the hypogastric plexus. This inhibits the detrusor and activates the internal urethral sphincter, promoting urine storage.
Somatic innervation via the pudendal nerve (S2-S4) controls the external urethral sphincter, which is skeletal muscle. Sensory innervation from stretch receptors provides feedback about filling status.
Dysfunction in any pathway can cause overactive bladder, urinary retention, or neurogenic bladder. Healthcare professionals must understand these neural connections for clinical practice.
Bladder Function and the Micturition Reflex
The bladder stores urine and periodically empties it through a process called micturition. This involves coordinated muscle contractions and nervous system control.
Normal Bladder Capacity
Your bladder can hold 400 to 500 milliliters of urine with minimal pressure increase. It can stretch to accommodate 600 to 800 milliliters. This ability comes from the bladder wall's compliance and the transitional epithelium's plasticity.
How the Micturition Reflex Works
The micturition reflex is a spinal reflex coordinated by the pontine micturition center in the brainstem. As the bladder fills, stretch receptors send sensory signals through pelvic nerves to the spinal cord and brain.
When voiding is appropriate, descending signals from the pontine micturition center activate parasympathetic neurons. This triggers synchronized detrusor contraction while relaxing the internal urethral sphincter. Simultaneously, the external urethral sphincter relaxes through inhibition of somatic motor neurons.
Emptying Efficiency
This coordinated contraction empties 80 to 90 percent of bladder contents within 20 to 30 seconds. Normal voiding happens 4 to 8 times daily. Nighttime continence stays strong because reflex thresholds are higher and sympathetic tone increases during sleep. Residual urine normally measures less than 50 milliliters.
Anatomical variations or damage to any neural pathway component can cause voiding dysfunction. Understanding anatomy helps explain incontinence, frequency, urgency, and retention in clinical patients.
Clinical Correlations and Common Pathologies
Bladder anatomy directly connects to recognizing and managing clinical conditions. Knowing the structure helps predict disease presentation and treatment options.
Common Bladder Conditions
Urinary tract infections (UTIs) are the most common bladder pathology. Infection risk increases with anatomical factors like the shorter female urethra and proximity to the anus.
Bladder stones form due to urinary stasis or infection. These must be distinguished from kidney stones by their location inside the bladder.
Bladder cancer, usually transitional cell carcinoma, arises from the mucosal lining and may present with hematuria. The trigone's distinct embryology makes it a unique site for potential cancer.
Vesicoureteral reflux occurs when the ureterovesical junction becomes incompetent, allowing backward urine flow. This typically affects children.
Functional Disorders
Overactive bladder results from detrusor hyperactivity, while underactive bladder occurs with detrusor weakness or outlet obstruction.
Neurogenic bladder develops following spinal cord injury or disease affecting innervation. This requires thorough understanding of neural control mechanisms.
Bladder diverticula are outpouchings of mucosa through the muscular wall, often near the ureteric orifices.
Surgical Considerations
Surgical interventions including cystoscopy, catheterization, and cystectomy require precise anatomical knowledge. Suprapubic catheters are placed above the pubic symphysis in the extraperitoneal bladder to avoid entering the peritoneal cavity.
Recognizing these clinical applications reinforces why detailed anatomical knowledge matters for healthcare professionals.