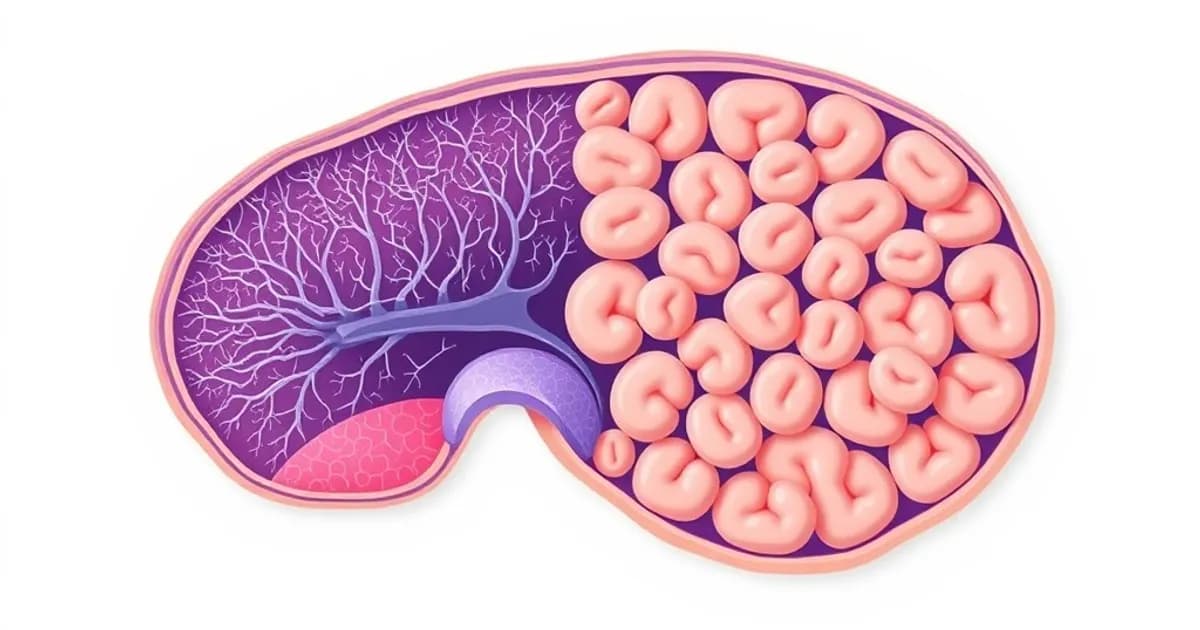
Olfactory Epithelium Structure and Organization
The olfactory epithelium is a specialized sensory tissue in the superior nasal cavity, covering approximately 10 square centimeters. It contains several distinct cell types arranged in layers as a pseudostratified columnar epithelium.
Cell Types and Layers
Olfactory receptor neurons (ORNs) are bipolar neurons with cell bodies in the epithelium. Their apical dendrite extends into the nasal mucus layer and contains specialized olfactory cilia. Their basal axon extends through the cribriform plate.
Supporting cells (sustentacular cells) are elongated columns extending from the apical surface to the basal lamina. They provide structural and metabolic support to ORNs throughout their lifespan.
Basal cells form a single layer against the basal lamina. These undifferentiated progenitor cells continuously generate new olfactory receptor neurons throughout life, a unique form of neurogenesis.
Supporting Structures
Bowman's glands are serous glands beneath the epithelium that secrete mucus. This mucus contains odorant-binding proteins that help dissolve odorant molecules.
The mucus layer is critical for olfactory function. It traps odorant molecules and allows them to reach the receptor cilia for detection.
Why This Architecture Matters
Each cell type contributes specifically to olfactory transduction and signal processing. Understanding how they work together reveals how smell functions at the cellular level.
Olfactory Receptor Neurons and Odorant Detection
Olfactory receptor neurons are unique because they directly contact the external environment. They continuously turn over throughout life, unlike most other neurons.
Receptor Expression and Specificity
Each ORN expresses only one type of olfactory receptor protein, which is a G-protein coupled receptor (GPCR). Humans have approximately 400 different types of olfactory receptors. The receptor expressed determines which odorants activate that neuron.
Signal Transduction Cascade
When an odorant binds to its receptor on the cilia, a specific sequence occurs:
- The receptor activates Golf, a G-protein
- Golf activates adenylyl cyclase, producing cyclic AMP
- Cyclic AMP opens cyclic nucleotide-gated ion channels
- Calcium and sodium enter the cilia, causing depolarization
- An action potential travels along the ORN axon
Journey to the Brain
ORN axons bundle together to form the olfactory nerve (cranial nerve I). This is the only cranial nerve that truly originates from peripheral neurons. Axons pass through the cribriform plate of the ethmoid bone to enter the olfactory bulb.
Continuous Renewal and Plasticity
ORNs are replaced every 30 to 40 days. This high turnover rate is unique among sensory neurons and enables neural plasticity and adaptation. It also fascinates regenerative medicine researchers studying how neurons regenerate.
Olfactory Bulb Organization and Layered Architecture
The olfactory bulb processes olfactory information first in the central nervous system. Located on the ventral frontal lobe, just above the cribriform plate, it has a highly organized layered structure.
The Five Main Layers
Olfactory nerve layer is the outermost layer containing unmyelinated ORN axons arriving from the epithelium.
Glomerular layer contains roughly 1,800 spherical structures called glomeruli in humans. ORNs expressing the same receptor type converge onto the same glomerulus. This convergence creates a topographic map of odorant receptors.
Mitral cell layer contains the soma of mitral cells, the principal output neurons. Their apical dendrites extend into single glomeruli. Their lateral dendrites extend throughout the external plexiform layer.
External plexiform layer contains synaptic connections between mitral cells, tufted cells, and granule cells. Lateral inhibition and signal modulation occur here.
Internal plexiform layer and granule cell layer follow, with granule cells providing feedback inhibition to mitral cells via reciprocal synapses.
Output Pathway
The axons of mitral and tufted cells form the lateral olfactory tract. This tract carries refined olfactory information to higher brain centers like the olfactory cortex and amygdala.
Glomerular Organization
Each glomerulus receives input from ORNs expressing the same receptor. This means all neurons responsive to a particular odorant converge onto the same glomerulus. Different odorants activate different spatial patterns of glomeruli.
Neural Circuits and Signal Processing in the Olfactory Bulb
The olfactory bulb contains sophisticated neural circuits that process olfactory information before it reaches the cortex. Beyond simple anatomy, these circuits refine signals through multiple mechanisms.
Primary Input Pathway
The basic circuit involves olfactory receptor neuron axons synapsing with mitral cell dendrites in glomeruli. This establishes a clear input-output pathway from periphery to brain.
Interneurons and Local Processing
Periglomerular cells surround individual glomeruli and receive input from ORNs. They provide local feedback within and between adjacent glomeruli. These cells sharpen olfactory discrimination by enhancing differences between similar odors.
Granule cells form reciprocal synapses with mitral cell lateral dendrites. These synapses are unusual because they lack a conventional postsynaptic density and involve both chemical and electrical coupling.
Top-Down Modulation
Granule cells receive centrifugal input (input from higher brain regions). This includes signals from the olfactory cortex, anterior olfactory nucleus, and amygdala. This top-down modulation is important for olfactory learning and attention.
Odor Maps and Refinement
The spatial activation pattern across glomeruli is called the odor map. This map is continually refined by the neural circuitry. Lateral inhibition mediated by granule cells sharpens these maps. Feedback from higher centers allows experience-dependent plasticity. Understanding these circuits reveals how the bulb transforms simple receptor activation into meaningful olfactory perception and memory formation.
Clinical Significance and Study Tips for Mastery
Understanding olfactory anatomy has important clinical applications. Anosmia (loss of smell) can result from epithelial damage or central lesions. Olfactory dysfunction is now recognized as an early symptom of Parkinson's disease and Alzheimer's disease.
Clinical Conditions
Viral infections can damage the olfactory epithelium and cause persistent anosmia. Traumatic head injury can shear olfactory axons at the cribriform plate. Olfactory bulb tumors or lesions can cause anosmia. Toxic chemical exposures can injure the epithelium.
Effective Study Strategies
Create visual mind maps showing the layered structure of the olfactory bulb and connections between layers. Then make flashcards for each layer with structure names and functions.
Use color-coded flashcards to distinguish between three epithelial cell types: receptor neurons, supporting cells, and basal cells.
Build sequence flashcards tracing the olfactory signal pathway. Start with odorant binding, move through the epithelium, along the olfactory nerve, through glomeruli, and to output neurons.
Make comparison flashcards contrasting olfactory receptors with other sensory receptor types. This highlights unique features of the olfactory system.
Use clinical case flashcards linking anatomical structures to disease states. This builds clinical thinking and retention.
Why Flashcards Excel Here
Flashcards combat dense terminology through spaced repetition. They consolidate spatial relationships between structures. Active recall creates strong memory traces, exactly what this complex topic demands for exam success.