The Four Phases of Fracture Healing and Callus Formation
Fracture healing occurs in four overlapping phases. Callus formation is central to the middle phases and drives bone repair.
Phase 1: Immediate Hematoma Formation
Within hours of injury, blood vessels rupture and create a fibrin clot at the fracture site. This hematoma seals the wound and establishes an inflammatory environment for healing to begin.
Phase 2: Inflammatory Phase
Within hours to days, immune cells migrate to the site. Neutrophils and macrophages clear debris and release signaling molecules that stimulate repair processes. This phase is essential for preparing the tissue for callus formation.
Phase 3: Soft Callus Formation
Days to weeks after injury, osteoblasts and fibroblasts proliferate and lay down a matrix of cartilage and woven bone. This soft callus is visible on X-rays but mechanically weak compared to mature bone. It provides initial stability while lacking the strength of mature bone.
Phase 4: Hard Callus and Remodeling
The soft callus undergoes endochondral ossification, transforming cartilage into woven bone. This phase takes weeks to months and represents transformation into stronger bone. Eventually, woven bone remodels into lamellar bone, the final mature form.
Recognizing where a fracture is in these phases helps predict healing outcomes and identify complications like nonunion (failure to bridge) or delayed union (slow progress).
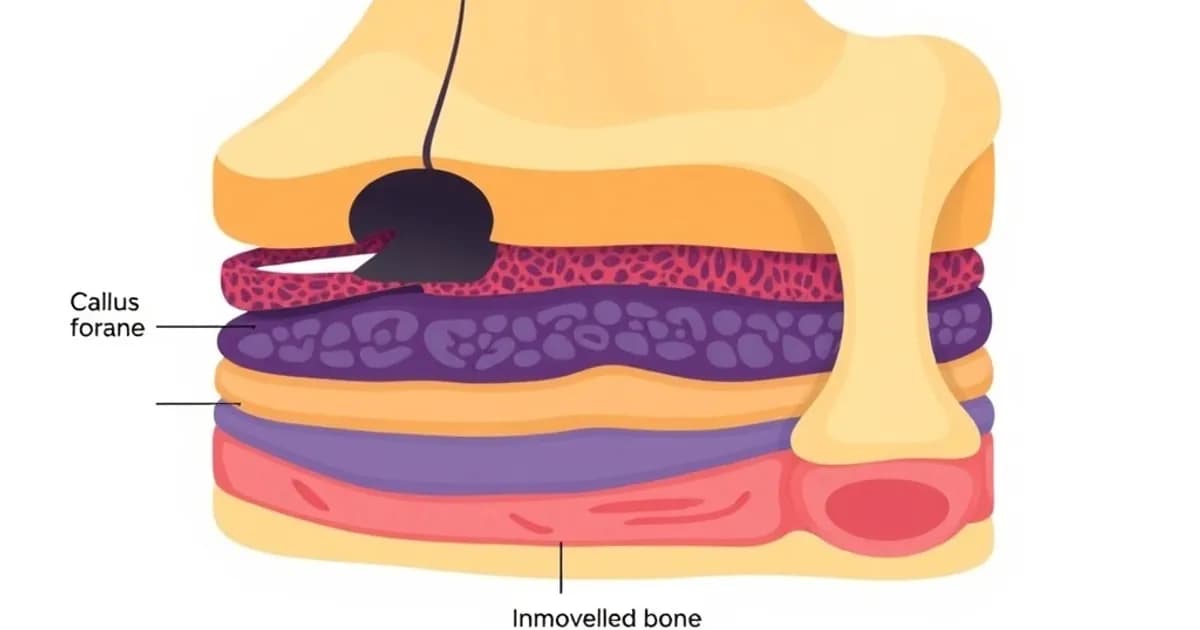
Histological Composition and Types of Callus
The callus develops in distinct locations and changes histologically as it matures. Location and composition determine its mechanical strength and healing timeline.
External vs. Internal Callus
External callus forms in surrounding soft tissues outside the bone. Internal callus develops within the medullary canal and surrounding bone. Both contribute to bridging the fracture.
Early Soft Callus Structure
Early soft callus consists mainly of fibrocartilage with randomly oriented collagen fibers. It provides some flexibility but limited strength. The peripheral regions may remain cartilaginous longer, providing shock absorption during healing.
Mature Hard Callus Structure
As callus matures, endochondral ossification converts cartilage into woven bone with random collagen orientation and higher mineralization. The final stage produces lamellar bone with parallel collagen fibers, providing superior strength and durability.
Continuous Remodeling
Importantly, the callus is not permanent. It undergoes continuous remodeling through targeted resorption and replacement, gradually transforming into normal cortical bone with Haversian systems. This remodeling process adapts bone structure to mechanical demands.
The rate and quality of histological progression depend on age, nutritional status, infection, fracture stability, and blood supply. On X-rays, soft callus appears hazy and ill-defined, while hard callus shows distinct bridging across the fracture line.
Molecular Factors and Growth Signaling in Callus Development
Multiple growth factors and signaling molecules orchestrate callus formation through coordinated cellular responses. These molecular signals explain why certain conditions improve or impair healing.
Bone Morphogenetic Proteins (BMPs)
BMPs, particularly BMP-2, BMP-6, and BMP-9, are powerful osteoinductive factors. They recruit mesenchymal stem cells and differentiate them into osteoblasts (bone-forming cells). These proteins are released from the bone matrix during fracture.
Angiogenic and Growth Factors
Fibroblast growth factors (FGFs) promote blood vessel formation and regulate early callus formation. Vascular endothelial growth factor (VEGF) is essential for building the blood vessel network that supplies oxygen and nutrients to developing callus.
Inflammatory and Regulatory Signals
Transforming growth factor-beta (TGF-β) stimulates cartilage formation in the soft callus phase. Interleukins and tumor necrosis factor-alpha (TNF-α) drive the initial inflammatory response that primes tissue for repair.
Hormone Support for Bone Formation
Parathyroid hormone (PTH) and insulin-like growth factors (IGFs) enhance bone formation. These pathways explain why calcium, vitamin D, and protein nutrition are critical for optimal healing.
Disruption of any pathway through infection, poor nutrition, smoking, or certain medications impairs callus formation. Understanding these mechanisms has led to clinical applications like recombinant BMP proteins to accelerate healing in critical fractures.
Factors Affecting Callus Formation and Healing Rate
Numerous intrinsic and extrinsic factors influence the speed and quality of callus formation. Clinicians use these factors to predict healing outcomes.
Age and Metabolic Factors
Children typically heal faster than adults due to greater metabolic activity and better blood supply. Elderly patients may have delayed healing due to reduced bone turnover and comorbidities. Endocrine status, particularly thyroid and parathyroid hormones, also influences healing significantly.
Nutritional Requirements
Adequate protein, calcium, vitamin C, and vitamin D directly impact callus formation. These nutrients are substrates for bone matrix synthesis and mineralization. Deficiencies slow callus development substantially.
Fracture Type and Stability
Simple, aligned fractures heal faster with minimal callus. Comminuted fractures (fractures with multiple fragments) with significant bone loss require larger, more robust callus formation. Immobilization and fracture stability are critical. Excessive movement at the fracture site inhibits callus formation and promotes fibrocartilage rather than bone.
Vascular Supply
Blood supply is perhaps the most critical factor for healing. Fractures with compromised blood flow (such as femoral neck fractures or crush injuries) heal poorly or not at all. Callus formation requires constant oxygen and nutrient delivery.
Lifestyle and Systemic Factors
Smoking reduces blood flow through vasoconstriction and impairs osteoblast function. Alcohol abuse and medications like corticosteroids suppress callus formation. Infection perpetuates inflammation and impairs osteoblast function. Understanding these factors allows prediction of healing outcomes and identification of at-risk patients.
Clinical Applications and Complications of Abnormal Callus Formation
Abnormalities in callus formation lead to important clinical complications. Recognizing these complications is essential for clinical practice.
Healing Complications: Delayed Union and Nonunion
Delayed union occurs when healing progresses slower than expected. Causes include inadequate immobilization, poor blood supply, or nutritional deficiencies. Nonunion is complete failure of callus to bridge the fracture within expected timeframes, resulting in a false joint called pseudarthrosis.
Types of Nonunion
Hypertrophic nonunion occurs when abundant callus forms but fails to mineralize into bone, often from excessive motion at the site. Atrophic nonunion involves minimal callus formation, typically from severe vascular compromise or infection. Both require different clinical interventions.
Other Callus Complications
Malunion results when the callus heals but fracture fragments are misaligned, causing functional or cosmetic problems. Exuberant callus is excessive but generally benign. Heterotopic ossification involves callus formation in abnormal locations, often seen after high-energy trauma.
Clinical Management Strategies
Optimize nutrition, ensure adequate immobilization, promote weight-bearing when appropriate to stimulate callus formation, and treat infections. In severe cases, use growth factor therapies or surgical interventions like bone grafting. Recognizing the healing phase and callus quality on imaging is essential for determining when to intervene versus allow natural healing.