Gross Anatomy of the Collecting Duct System
The collecting duct system begins where multiple distal convoluted tubules converge in the renal cortex. These initial ducts are 20-30 micrometers in diameter and descend through the renal medulla, progressively merging into larger ducts.
Hierarchical Organization
The system follows a clear structural progression:
- Cortical collecting ducts branch into medullary collecting ducts
- Medullary ducts converge into papillary ducts (ducts of Bellini) in the renal papilla
- Collecting ducts course through the medulla in parallel arrangement, creating medullary rays
Epithelial Transitions
The lining transitions from simple cuboidal epithelium in cortical ducts to columnar epithelium in larger medullary and papillary ducts. Collecting ducts are embedded in the interstitium, positioned close to the vasa recta. This capillary network exchanges solutes and water with tubular fluid.
Functional Significance
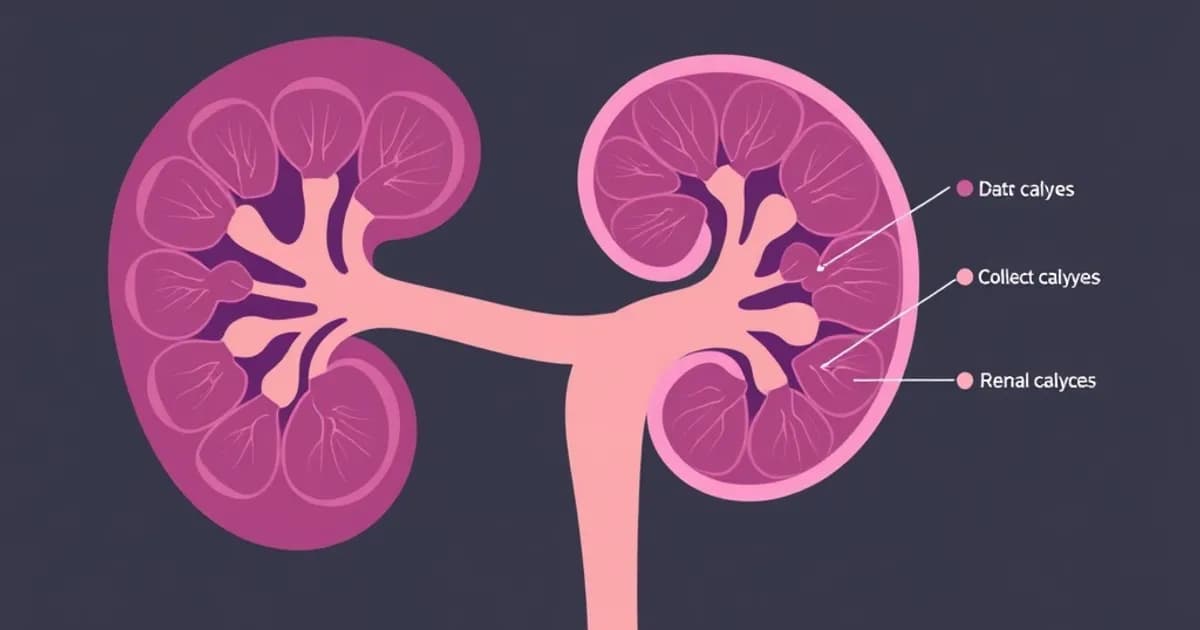
The papillary ducts terminate at the area cribrosa, where urine exits into the minor calyces. Despite representing only 5 percent of total tubule length, the collecting duct system disproportionately determines final urine composition and volume.
Cellular Composition and Histology
The collecting duct epithelium contains two primary cell types: principal cells and intercalated cells. Each type has distinct ultrastructural features and functions.
Principal Cells
Principal cells comprise approximately 60 percent of the epithelial lining. They are cuboidal cells with relatively few mitochondria and microvilli. These cells express aquaporin-2 water channels on their apical membrane, inserted in response to antidiuretic hormone (ADH).
The basolateral membrane contains aquaporin-3 and aquaporin-4, allowing water to exit into the interstitium. Principal cells also possess epithelial sodium channels (ENaC) on their apical membrane, enabling sodium reabsorption that creates osmotic gradients for water reabsorption.
Intercalated Cells
Intercalated cells comprise the remaining epithelium and appear darker due to abundant mitochondria. Two subtypes exist:
- Alpha-intercalated cells secrete acid through apical H-ATPase pumps
- Beta-intercalated cells secrete bicarbonate through basolateral chloride-bicarbonate exchangers
Functional Implications
Mitochondrial abundance in intercalated cells reflects their high metabolic activity for active transport. The distinct membrane transporters in each cell type explain how the collecting duct performs multiple regulatory functions simultaneously.
Epithelial Characteristics and Permeability Properties
The collecting duct epithelium is classified as simple epithelium with distinctive permeability characteristics. These properties change along the tubule's length and respond to hormonal signals.
Water Permeability Regulation
In the cortical collecting duct, the epithelium is relatively impermeable to water without ADH. This allows urine concentration to increase as the tubule descends into the hyperosmolar medulla. When ADH binds to V2 receptors on principal cells, it triggers insertion of aquaporin-2 channels into the apical membrane.
This transformation converts the epithelium into a water-permeable membrane. The regulated response allows the body to produce either concentrated or dilute urine depending on hydration status.
Ion and Urea Transport
The collecting duct has low sodium permeability in most segments. Sodium reabsorption occurs only through active transport via ENaC channels on principal cells. However, the inner medullary collecting duct shows increased permeability to urea, allowing passive reabsorption that contributes to medullary osmolarity and the countercurrent multiplier system.
Tight Junction Architecture
Epithelial tight junctions are relatively tight, preventing passive paracellular flow. This ensures transport is transcellular and can be tightly regulated. The transition from water-impermeable to water-permeable epithelium is one of the most testable aspects of kidney function regulation.
Functional Anatomy and Hormonal Regulation
The collecting duct system's function is intimately linked to hormonal signals, particularly ADH and aldosterone.
Antidiuretic Hormone Mechanism
ADH acts on principal cells through V2 vasopressin receptors coupled to G-protein signaling. This increases intracellular cAMP, which activates protein kinase A. The enzyme phosphorylates aquaporin-2, leading to recruitment of water channel vesicles to the apical membrane.
This mechanism allows water reabsorption to be rapidly turned on or off. The body can produce concentrated urine when water conservation is needed or dilute urine when excess water must be excreted.
Aldosterone Function
Aldosterone acts on principal cells to increase epithelial sodium channel expression and Na-K-ATPase activity. This promotes sodium reabsorption and potassium secretion. Mineralocorticoid receptor signaling increases gene transcription for sodium transport machinery.
Acid-Base Regulation
Intercalated cells respond to acid-base status through various signaling mechanisms. They adjust hydrogen ion secretion or bicarbonate secretion as needed. The collecting duct's strategic location deep in the medulla positions it to respond to medullary osmolarity, signaling hydration status.
The presence of multiple receptor types and signaling pathways makes the collecting duct essential for integrating hormonal systems with kidney function.
Clinical Significance and Study Strategies for Mastery
Clinical pathologies demonstrate the importance of collecting duct structure and function. Understanding these conditions requires solid foundational knowledge of normal anatomy and physiology.
Common Clinical Conditions
- Nephrogenic diabetes insipidus: Mutations affect aquaporin-2 or V2 receptor genes, preventing urine concentration despite adequate ADH
- Bartter and Gitelman syndromes: Defects in ion transporters indirectly affect collecting duct function, causing electrolyte disturbances
- Renal tubular acidosis type 1 (RTA-1): Dysfunction of alpha-intercalated H-ATPase pumps prevents adequate acid secretion, causing hyperchloremic acidosis
Effective Flashcard Strategies
Create flashcards that link anatomical structures to functions. One side should describe a cellular feature (like principal cells containing aquaporin-2), while the reverse explains the functional consequence (water reabsorption in response to ADH).
Organize flashcards by region (cortical versus medullary collecting ducts) and by function (water reabsorption, sodium reabsorption, acid-base regulation). Include visual components that identify cell types by ultrastructural features, such as mitochondrial density and membrane characteristics.
Active Recall Practice
Practice clinical scenarios where patients present with abnormal urine osmolarity or electrolyte imbalances. Challenge yourself to explain the underlying anatomical disruption. Create comparison flashcards contrasting principal and intercalated cells, or comparing cortical and medullary collecting duct characteristics.
Review electrolyte regulation simultaneously with structural features, as the two are inseparable. Use mnemonics for remembering which intercalated cell type performs which function. Regular active recall testing through flashcard review substantially improves retention compared to passive reading.