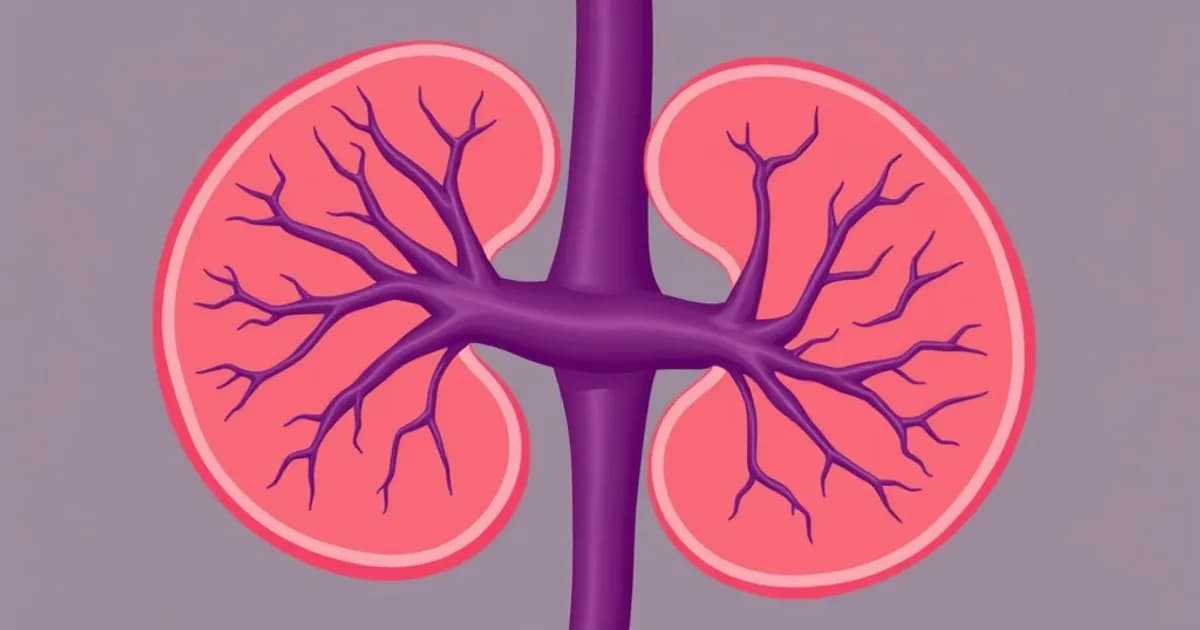
Gross Renal Arterial Supply and Origin
Main Renal Artery Origin and Course
The renal arteries originate from the lateral abdominal aorta at the level of the L1-L2 vertebra. This location sits just below the superior mesenteric artery. Each kidney typically receives one main renal artery, though anatomical variations are common.
Accessory renal arteries occur in approximately 25-30% of the population. The right renal artery arises more inferiorly and passes posterior to the inferior vena cava. The left renal artery has a more horizontal course across the midline.
Renal Artery Characteristics
Each renal artery measures about 4-5 centimeters before entering the renal hilum. The renal artery is unique because it does not supply the gastrointestinal tract, unlike other abdominal aorta branches.
Upon entering the kidney at the hilum, the renal artery divides into segmental arteries. These arteries supply distinct regions of the kidney and have minimal collateral circulation.
Clinical Significance of Renal Artery Anatomy
Renal artery stenosis, thrombosis, or dissection can cause significant kidney damage due to the lack of collateral blood supply. The renal vein typically crosses anterior to the renal artery at the hilum, which matters during surgery and imaging interpretation.
Surgeons and interventional radiologists must understand these relationships when planning procedures. Any vascular injury during these interventions can have serious consequences for kidney function.
Microvascular Architecture and Afferent-Efferent Arterioles
Arteriole Branching Pattern
After segmental arteries branch, the renal circulation progressively narrows. Blood flows through interlobar arteries, arcuate arteries, and finally interlobular arteries that supply individual nephrons.
Each interlobular artery gives rise to multiple afferent arterioles. Each afferent arteriole feeds a single glomerulus.
The Afferent-Efferent Arteriole Diameter Difference
The afferent arteriole is wider in diameter than the efferent arteriole. This diameter difference creates a critical pressure gradient for glomerular filtration. The arrangement allows approximately 20% of plasma entering the glomerulus to filter into Bowman's capsule.
This anatomical feature is physiologically regulated by:
- Renin-angiotensin-aldosterone system
- Local autoregulation mechanisms
- Sympathetic nervous system input
Fine-Tuning Filtration Pressure
Juxtaglomerular cells in the afferent arteriole walls sense blood pressure changes and regulate renin secretion. The efferent arteriole contains specialized smooth muscle cells that respond to angiotensin II.
This allows the kidney to fine-tune glomerular filtration pressure based on systemic needs. In diabetic nephropathy, both afferent and efferent arterioles are affected, but at different disease stages.
The glomerular capillary network itself has specialized fenestrated endothelium. The filtration barrier consists of three layers: endothelium, basement membrane, and podocytes. All depend on healthy afferent arteriole supply.
Peritubular Capillaries and Vasa Recta
Efferent Arteriole Branching and Two Capillary Systems
The efferent arteriole does not drain directly into veins. Instead, it branches into two distinct capillary systems depending on nephron location.
Cortical nephrons comprise approximately 85% of all nephrons. Their efferent arterioles branch into peritubular capillaries surrounding the proximal and distal convoluted tubules and cortical collecting duct.
Peritubular Capillaries and Reabsorption
Peritubular capillaries enable reabsorption of useful substances back into the bloodstream. These substances include glucose, amino acids, ions, and water. The hydrostatic pressure in peritubular capillaries is lower than in glomerular capillaries, promoting reabsorption rather than filtration.
This pressure difference is essential for the kidney's ability to conserve valuable solutes and fluid.
Vasa Recta and Urine Concentration
Juxtamedullary nephrons have efferent arterioles extending into the medulla. These form the vasa recta, which are specialized hairpin-shaped capillaries descending alongside the loop of Henle.
The vasa recta create a countercurrent multiplier system that concentrates urine and conserves water. These vessels have thin walls and slow blood flow, permitting efficient solute and water exchange.
The vasa recta are essential for producing concentrated urine, a critical adaptation for water conservation. Understanding this distinction between peritubular capillaries and vasa recta explains kidney function during dehydration and heart failure.
Renal Venous Drainage and Clinical Variations
Venous Drainage Pattern
Blood exits the kidney through venous channels that mirror the arterial supply in reverse order. Peritubular capillaries and vasa recta drain into interlobular veins. These coalesce into arcuate veins and interlobar veins, eventually forming the renal vein.
The renal veins are larger in diameter than their corresponding arteries. They also have thinner walls, allowing easier distension.
Left and Right Renal Vein Differences
The left renal vein is typically longer than the right renal vein. It must cross the midline to drain into the inferior vena cava. The left renal vein commonly receives the left gonadal vein, left adrenal vein, and left phrenic vein.
The right renal vein is shorter and drains more directly into the inferior vena cava. This anatomical asymmetry has important surgical implications.
Clinical Variations and Complications
Renal vein thrombosis is a serious condition occurring with nephrotic syndrome, hypercoagulable states, or trauma. It can result in acute kidney dysfunction or chronic renal atrophy.
Anatomical variations in venous drainage are less common than in arterial supply. However, multiple renal veins or retroaortic renal veins can occur. The retroaortic left renal vein, present in approximately 5% of the population, crosses behind the aorta rather than in front.
Understanding venous anatomy is essential for interventional radiologists performing renal vein catheterization and for surgeons planning nephrectomy or renal transplantation.
Clinical Correlates and Pathophysiology of Vascular Disease
Renal Artery Stenosis and Hypertension
Renal vascular disease has significant clinical implications affecting kidney function and systemic blood pressure. Renal artery stenosis is commonly caused by atherosclerotic disease in older patients or fibromuscular dysplasia in younger patients.
Stenosis reduces blood flow to the kidney and activates the renin-angiotensin-aldosterone system, leading to secondary hypertension. Diagnostic tools include duplex ultrasound, CT angiography, and MR angiography.
Acute Vascular Emergencies
Acute renal artery dissection presents with sudden flank pain and elevated creatinine. Renal infarction, caused by thromboembolic phenomena or dissection, results in tissue necrosis and can be difficult to diagnose clinically.
Diabetic Kidney Disease
Diabetic nephropathy begins with selective efferent arteriole injury due to hyperglycemia-induced angiotensin II activation. This causes glomerular hyperfiltration followed by progressive glomerular damage.
ACE inhibitors and angiotensin receptor blockers can slow disease progression by dilating the efferent arteriole.
Acute Kidney Injury and Other Vascular Conditions
Acute kidney injury can result from prerenal causes such as renal hypoperfusion during shock or dehydration. In these cases, the kidney is structurally intact but receives insufficient blood flow.
Contrast-induced nephropathy occurs when radiocontrast agents cause arteriolar vasoconstriction and direct tubular toxicity in susceptible patients.
Clinical Decision Making
Understanding vascular anatomy allows clinicians to predict disease outcomes. Stenosis of a single renal artery may not significantly reduce total kidney function because the contralateral kidney compensates. However, bilateral stenosis can cause progressive chronic kidney disease.
Renal transplant recipients require vascular anastomoses carefully placed to ensure adequate graft blood supply. This makes understanding renal vascular anatomy essential for transplant surgeons.